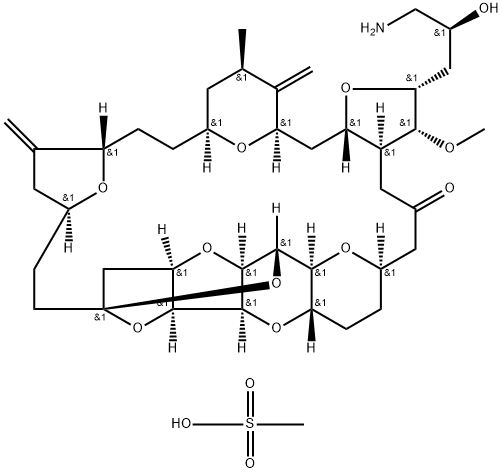
441045-17-6
| Name | Eribulin Mesylate |
| CAS | 441045-17-6 |
| EINECS(EC#) | 813-106-4 |
| Molecular Formula | C40H59NO11.CH4O3S |
| MDL Number | MFCD22572760 |
| Molecular Weight | 826.01 |
| MOL File | 441045-17-6.mol |
Synonyms
Halaven
B1939 mesylate
E7389 mesylate
Eribulin Mesylate
Eribulin Mesilate
ER-086526 mesylate
Eribulin mesylate
Halaven
EribulinMesylate USP/EP/BP
Chemical Properties
| storage temp. | Store at -20°C, protect from light, stored under nitrogen |
| solubility | Soluble in DMSO |
| form | Powder |
| InChIKey | QAMYWGZHLCQOOJ-PEKQNERQNA-N |
Hazard Information
Uses
Eribulin Mesylate is a synthetic analog of the marine natural product halichondrin B.
Definition
ChEBI: A methanesulfonate salt obtained by reaction of eribulin with one equivalent of methanesulfonic acid. A fully synthetic macrocyclic ketone analogue of marine sponge natural products. Inhibits growth phase of microtubules via tubulin-based antimitotic mecha
ism, which leads to G2/M cell-cycle block, disruption of mitotic spindles, and, ultimately, apoptotic cell death after prolonged mitotic blockage.
Biological Activity
eribulin mesylate (e7389 mesylate), a synthetic analogue of halichondrin b in phase iii
Clinical Use
Eribulin is a highly potent cytotoxic agent approved in the U.S. for the treatment of metastatic breast
cancer for patients who have received at least two previous chemotherapeutic regimens. Eribulin
was discovered and developed by Eisai and it is currently undergoing clinical evaluation for the
treatment of sarcoma (PhIII) and non-small cell lung cancer which shows progression after platinumbased
chemotherapy and for the treatment of prostate cancer (PhII). Early stage clinical trials are also
underway to evaluate eribulin’s efficacy against a number of additional cancers. Eribulin is a structural analog of the marine natural product halichondrin B. Its mechanism of action involves the disruption of
mitotic spindle formation and inhibition of tubulin polymerization which results in the induction of cell
cycle blockade in the G2/M phase and apoptosis.
Synthesis
Several synthetic routes for the preparation of
eribulin have been disclosed, each of which utilizes the same strategy described by Kishi and
co-workers for the total synthesis of halichondrin B. Although the scales of these routes were not
disclosed in all cases, this review attempts to highlight what appears to be the production-scale route
based on patent literature. Nonetheless, the synthesis of eribulin represents a significant
accomplishment in the field of total synthesis and brings a novel chemotherapeutic option to cancer
patients.
The strategy to prepare eribulin mesylate (V) employs a convergent synthesis featuring the following: the late stage coupling of sulfone 22 and aldehylde 23 followed by macrocyclization under Nozaki-Hiyami-Kishi coupling conditions, formation of a challenging cyclic ketal, and installation of the primary amine. Sulfone 22 was further simplified to aldehyde 24 and vinyl triflate 25 which were coupled through a Nozaki-Hiyami-Kishi reaction.
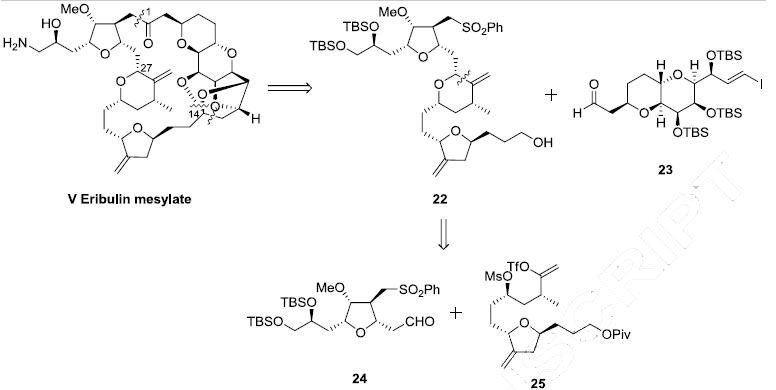
With the three key fragments 23, 24 and 25 along with the entire molecule completed, the next step was to assemble them and complete the synthesis of eribulin. Aldehyde 24 was coupled to vinyl triflate 25 using an asymmetric Nozaki- Hiyami-Kishi reaction using CrCl2, NiCl2, Et3N and chiral ligand 67 (the Scheme) to give alcohol 83 (Scheme 11). Formation of the THP ring was accomplished by reaction with KHMDS which allowed for displacement of the mesylate with the secondary alcohol and provided the THP containing product in 72% yield for the three steps. The pivalate ester group was removed with DIBAL-H to give the western fragment 22 in 92% yield.
The completion of the synthesis of eribulin is illustrated in Scheme 12. The lithium anion of sulfone 22 generated upon reaction with nBuLi was coupled to aldehyde 23 to give diol 84 in 84% yield. Both of the alcohol functional groups of 84 were oxidized using a Dess-Martin oxidation in 90% yield and the resulting sulfone was removed via a reductive cleavage upon reaction with SmI2 to give ketoaldehyde 85 in 85% yield. Macrocyclization of 85 was accomplished via an asymmetric Nozaki Hiyami-Kishi reaction using CrCl2, NiCl2, Et3N and chiral ligand 67 to give alcohol 86 in 70% yield.
Modified Swern oxidation of the alcohol provided the corresponding ketone in 91% yield and this was followed by removal of the five silyl ether protecting groups upon reaction with TBAF and subsequent cyclization to provide ketone 87. Compound 87 was treated with PPTS to provide the “caged” cyclic ketal 88 in 79% over two steps. The vicinal diol of 88 was reacted with Ts2O in collidine to affect selective tosylation of the primary alcohol and this crude product was reacted with ammonium hydroxide to install the primary amine to give eribulin which was treated with methanesulfonic acid in aqueous ammonium hydroxide to give eribulin mesylate (V) in 84% yield over the final 3 steps.

The strategy to prepare eribulin mesylate (V) employs a convergent synthesis featuring the following: the late stage coupling of sulfone 22 and aldehylde 23 followed by macrocyclization under Nozaki-Hiyami-Kishi coupling conditions, formation of a challenging cyclic ketal, and installation of the primary amine. Sulfone 22 was further simplified to aldehyde 24 and vinyl triflate 25 which were coupled through a Nozaki-Hiyami-Kishi reaction.

With the three key fragments 23, 24 and 25 along with the entire molecule completed, the next step was to assemble them and complete the synthesis of eribulin. Aldehyde 24 was coupled to vinyl triflate 25 using an asymmetric Nozaki- Hiyami-Kishi reaction using CrCl2, NiCl2, Et3N and chiral ligand 67 (the Scheme) to give alcohol 83 (Scheme 11). Formation of the THP ring was accomplished by reaction with KHMDS which allowed for displacement of the mesylate with the secondary alcohol and provided the THP containing product in 72% yield for the three steps. The pivalate ester group was removed with DIBAL-H to give the western fragment 22 in 92% yield.
The completion of the synthesis of eribulin is illustrated in Scheme 12. The lithium anion of sulfone 22 generated upon reaction with nBuLi was coupled to aldehyde 23 to give diol 84 in 84% yield. Both of the alcohol functional groups of 84 were oxidized using a Dess-Martin oxidation in 90% yield and the resulting sulfone was removed via a reductive cleavage upon reaction with SmI2 to give ketoaldehyde 85 in 85% yield. Macrocyclization of 85 was accomplished via an asymmetric Nozaki Hiyami-Kishi reaction using CrCl2, NiCl2, Et3N and chiral ligand 67 to give alcohol 86 in 70% yield.
Modified Swern oxidation of the alcohol provided the corresponding ketone in 91% yield and this was followed by removal of the five silyl ether protecting groups upon reaction with TBAF and subsequent cyclization to provide ketone 87. Compound 87 was treated with PPTS to provide the “caged” cyclic ketal 88 in 79% over two steps. The vicinal diol of 88 was reacted with Ts2O in collidine to affect selective tosylation of the primary alcohol and this crude product was reacted with ammonium hydroxide to install the primary amine to give eribulin which was treated with methanesulfonic acid in aqueous ammonium hydroxide to give eribulin mesylate (V) in 84% yield over the final 3 steps.

storage
Store at -20°C, protect from light, stored under nitrogen
Mode of action
Eribulin Mesylate is the mesylate salt of a synthetic analogue of halichondrin B, a substance derived from a marine sponge (Lissodendoryx sp.) with antineoplastic activity. Eribulin binds to the vinca domain of tubulin and inhibits the polymerization of tubulin and the assembly of microtubules, resulting in inhibition of mitotic spindle assembly, induction of cell cycle arrest at G2/M phase, and, potentially, tumor regression.
References
Eribulin mesylate: a novel halichondrin B analogue for the treatment of metastatic breast cancer DOI: 10.2146/ajhp110237
From micrograms to grams: scale-up synthesis of eribulin mesylate DOI: 10.1039/c3np70051h
Eribulin mesylate: mechanism of action of a unique microtubule-targeting agent DOI: 10.1158/1078-0432.CCR-14-3252
From micrograms to grams: scale-up synthesis of eribulin mesylate DOI: 10.1039/c3np70051h
Eribulin mesylate: mechanism of action of a unique microtubule-targeting agent DOI: 10.1158/1078-0432.CCR-14-3252
Supplier
Hubei Zhongshan Medical Technology Co., Ltd
Telephone027-61907345 13397111514
Websitehttps://www.chemicalbook.com/supplier/23792894/
WuYan Pharmaceutical Technology (Shanghai) Co., Ltd.
Telephone021-61001086 13671938079
Websitehttp://www.wuyanpharm.com
Hefei Hirisun Pharmatech Co., Ltd.
Telephone+86-0551-62678551 +86-15056975894
Websitehttp://www.hirisunpharm.com
Wuhan Wiseman Bioengineering Co.,Ltd
Telephone027-59506022 13419526507
Websitehttp://www.whwiseman.com
Abydos Scientific
Telephone025-84767922 18936879710
Websitehttp://www.abydoscientific.com/index.html
Haoyuan Chemexpress Co., Ltd.
Telephone021-58950125
Websitehttps://www.chemexpress.com.cn/
Nanjing Becas pharmatech Ltd.
Telephone 13912903754
Website
Chemvon Biotechnology Co., Ltd
Telephone021-50790412
Websitehttp://www.chemvon.com
Chembest Research Laboratories Limited
Telephone021-20908456
Websitehttp://www.BioChemBest.com
Energy Chemical
Telephone021-021-58432009 400-005-6266
Websitehttp://www.energy-chemical.com
Shanghai Fuhe Chemistry Technology Co., Ltd.
Telephone0086-21-67651709
Websitehttps://www.chemicalbook.com/ShowSupplierProductsList15402/0.htm
NCE Biomedical Co.,Ltd.
Telephone4000-027-021 |24 +86-13986109188 | +86-15623472865 | +81-08033611988
Websitehttps://www.chemicalbook.com/ShowSupplierProductsList15748/0.htm
Haoyuan Chemexpress Co., Ltd.
Telephone021-58950125
Websitehttp://gb.chemexpress.com.cn
China Nobel Chem Co., Limited
Telephone+86(0)21 60484900
Websitehttps://www.chemicalbook.com/ShowSupplierProductsList15874/0.htm
Shanghai Witofly Chemical Co.,Ltd
Telephone
Websitehttp://www.witofly.com
TargetMol Chemicals Inc.
Telephone021-021-33632979 15002134094
Websitehttps://www.targetmol.cn/
Wuxi Zhongkun Biochemical Technology Co., Ltd.
Telephone0510-85629785 18013409632
Websitehttp://www.reading-chemicals.com
BrightGene Bio-medical Technology Co., Ltd.
Telephone0512-0512-62551764 18013103761
Websitehttp://www.bright-gene.com
1of4
PROMPT×
PROMPT
The What'sApp is temporarily not supported in mainland China
The What'sApp is temporarily not supported in mainland China
Cancel
Determine