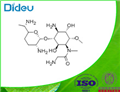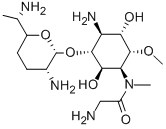
FORTIMICIN
- Product NameFORTIMICIN
- CAS55779-06-1
- CBNumberCB7116881
- MFC17H35N5O6
- MW405.49
- MDL NumberMFCD00864879
- MOL File55779-06-1.mol
- MSDS FileSDS
Chemical Properties
| Melting point | >200° (dec) |
| alpha | D25 +87.5° (c = 0.1 in water) |
| Boiling point | 526.82°C (rough estimate) |
| Density | 1.0897 (rough estimate) |
| refractive index | 1.7600 (estimate) |
| pka | 13.16±0.70(Predicted) |
| FDA UNII | 7JHD84H15J |
FORTIMICIN Chemical Properties,Usage,Production
Originator
Fortimicin sulfate,Youngjin PharmaDefinition
ChEBI: An amino cyclitol glycoside that is L-chiro-inositol in which the hydroxy groups at positions 1, 4, and 6 are replaced by aminoacetyl)methylamino, amino, and methoxy groups, respectively, and in which the hydroxy group at posi ion 3 is converted to the corresponding 2,6-diamino-2,3,4,6,7-pentadeoxy-beta-L-lyxo-heptopyranoside. The major component of fortimicin, obtained from Micromonospora olivasterospora. It is adm nistered (as the sulfate salt) by intramuscular injection or intravenous infusion for the treatment of severe systemic infections due to sensitive Gram-negative organisms.Manufacturing Process
Aminoglycoside antibiotic complex produced by Micromonospora used as a seed strain. One loopful of the seed strain is inoculated into 10 ml of a seed medium containing 2% glucose, 0.5% peptone, 0.5% yeast extract and 0.1% calcium carbonate (pH 7.5 before sterilization) in a 50 ml large test tube. Culturing is carried out at 30°C for 5 days. 10 ml of the seed culture broth is then inoculated into 30 ml of a second seed medium in a 250 ml Erlenmeyer flask. The composition of the second seed medium is the same as that of the first seed medium. The second seed culturing is carried out at 30°C for 2 days with shaking. Then 30 ml of the second seed culture broth is inoculated into 300 ml of a third seed medium in a 2 L Erlenmeyer flask provided with baffles. The composition of the third seed medium is the same as that of the first seed medium. The third seed culturing is carried out at 30°C for 2 days with shaking and 1.5 L of the third seed culture broth (corresponding to the content of five flasks) is inoculated into 15 L of a fourth seed medium in a 30 L glass jar fermenter. The composition of the fourth seed medium is the same as that of the first seed medium. Culturing in the jar fermenter is carried out at 37°C for 2 days with aeration and stirring (revolution: 350 r.p.m., aeration: 15 L/min). Thereafter, 15 L of the fourth seed culture broth is inoculated into 150 L of a main fermentation medium in a 300 L fermenter. The main fermentation medium comprises 4% soluble starch, 2% soybean meal, 1% corn steep liquor, 0.05% K2HPO4, 0.05% MgSO4·7H2O, 0.03% KCl and 0.1% CaCO3 (pH 7.5 before sterilization). Culturing in the fermenter is carried out at 37°C for 4 days with aeration and stirring (revolution: 150 r.p.m., aeration: 80 L/min).After the completion of culturing, the resulting fermentation broth is adjusted to a pH of 2.5 with concentrated sulfuric acid, and stirred for 30 minutes. Then, about 7 kg of a filter aid, Radiolite No. 600 (product of Showa Kagaku Kogyo Co., Ltd., Japan) is added thereto and the microbial cells are removed by filtration. The filtrate is adjusted to a pH of 7.5 with 6 N sodium hydroxide and passed through a column packed with about 20 L of a cation exchange resin, Amberlite IRC-50 (ammonium form), and the effluent is discarded. Active substances are adsorbed on the resin. After washing the resin with water, the adsorbed active substances are eluted out with 1 N aqueous ammonia. Activity of the eluate is determined by a paper disc method, using an agar plate of Bacillus subtilis No. 10707. The active fractions are collected and the mixture is concentrated to about 1 L under reduced pressure. The concentrate is passed through a column packed with 500 ml of an anion exchange resin, Dowex 1x2 (OH- form). Then, about 2 L of water is passed through the column, whereby impurities are removed and active substances are eluted out. The thus obtained active fractions are collected, and concentrated to about 100 ml under reduced pressure, and the resulting concentrate is passed through a column packed with about 50 ml of active carbon powder. The active substances are adsorbed onto the carbon powders. Then, the column is washed with water and the effluent and the washing water are discarded. Then, the adsorbed active substances are eluted out with 0.2 N sulfuric acid. Activity of the eluate is determined by the paper disc method using Bacillus subtilis, and the active fractions are collected. The thus obtained fractions are passed through a column of Dowex 44 (OH- form), and active substances are eluted out with water. The active fractions are again collected and concentrated to about 50 ml. The thus obtained concentrate is lyophilized, whereby about 32 g of a crude powder containing Fortimicin A is obtained. The crude powder exhibits an activity of 575 unit/mg (the activity of 1 mg of a pure product corresponds to 1000 units).
Then 10 g of the crude powder is placed as a thin and uniform layer on 500 ml of silica gel packed in a glass column. The glass column is prepared by suspending the silica gel in a solvent of the lower layer of a mixture comprising chloroform, isopropanol and 17% aqueous ammonia (2:1:1 by volume), and then packing the suspension tightly in the column as a uniform layer, and thereafter washing with the same solvent. After placing the crude powder at the head of the column, elution is carried out with the abovedescribed solvent by gradually pouring into the column from its top, and thereafter elution is carried out at a flow rate of about 50 ml/hour. The eluate is obtained as fractions of 20 ml each, and the activity of each fraction is determined by a paper disc method. Fortimicin B is eluted out at first. Thereafter, fractions containing Fortimicin A are obtained. The active fractions are subjected to paper chromatography, and the fractions containing Fortimicin A are collected and concentrated under reduced pressure to completely remove the solvent. The concentrate is then dissolved in a small amount of water. After freeze-drying the solution, about 1.8 g of purified preparate of the free base of Fortimicin A is obtained. The activity of the preparate is about 970 unit/mg. White amorphous powder, MP: >200° (dec.). [α] D 25 +87.5° (c = 0.1 in water), solves in water and lower alcohols, insoluble in organic solvents.
Therapeutic Function
AntibioticAntimicrobial activity
A pseudodisaccharide aminoglycoside produced by Micromonospora olivoasterospora. Formulated as the sulfate. Intrinsic activity is similar to that of amikacin for most groups of organisms, but activity against Ps. aeruginosa is relatively poor. It is resistant to many aminoglycoside-modifying enzymes, but is sensitive to AAC(3) and the APH(2″)/AAC(6′) bifunctional enzyme.Peak concentrations of 10–12 mg/L were found in the blood following 200 mg intravenous or intramuscular administration to volunteers. The plasma half-life was 1.5–2 h. Over 85% of the drug was recovered in urine during the 8 h following administration.
Toxicity and side effects are similar to those observed with other aminoglycosides. Where the drug is available it is used instead of amikacin in the treatment of infections caused by susceptible organisms.
Preparation Products And Raw materials
FORTIMICIN Supplier
Global(26)Suppliers
| Supplier | Tel | Country | ProdList | Advantage | |
|---|---|---|---|---|---|
| +86-29-87569265 +86-18612256290 |
1056@dideu.com | China | 3632 | 58 | |
| +86-029-89586680 +86-18192503167 |
1026@dideu.com | China | 9358 | 58 | |
| +86-29-89586680 +86-15129568250 |
1026@dideu.com | China | 29271 | 58 | |
| +86-057181025280; +8617767106207 |
sales@molcore.com | China | 49739 | 58 | |
| +1-781-999-5354 | support@targetmol.com | United States | 19973 | 58 | |
| +86-19164747840 +86-13119157289 |
13119157289@163.com | China | 2971 | 58 | |
| 0571-82693216 | info@yuhaochemical.com | China | 9394 | 52 | |
| 1-631-485-4226; 16314854226 |
info@bocsci.com | United States | 14059 | 65 | |
| 010-56205725 | waley188@sohu.com | China | 12338 | 58 | |
| 0512-56316828 | info@amateksci.com | China | 28822 | 58 |
55779-06-1, FORTIMICINRelated Search
PROMPT×
PROMPT
The What'sApp is temporarily not supported in mainland China
The What'sApp is temporarily not supported in mainland China
Cancel
Determine