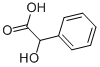
Mandelic acid turns brown when exposed to light. It can be esterified easily with alcohols in the presence of hydrogen chloride.
(R,S)-Mandelic acid (a-hydroxyphenylacetic acid, phenylglycolic acid), C6H5CH(OH)COOH, C8H8O3, Mr 152.15, mp 118 – 121℃, is a colorless crystalline powder. It is readily soluble in ethanol and diethyl ether, less soluble in chloroform, and insoluble in petroleum ether. Its water solubility is 15 g per 100 mL of water. In alkaline solution, mandelic acid dissolves to form salts.
ChEBI: Mandelic acid is a 2-hydroxy monocarboxylic acid that is acetic acid in which two of the methyl hydrogens are substituted by phenyl and hydroxyl groups. It has a role as an antibacterial agent and a human xenobiotic metabolite. It is a 2-hydroxy monocarboxylic acid and a member of benzenes. It is functionally related to an acetic acid. It is a conjugate acid of a mandelate.
(R,S)-mandelic acid is produced
by hydrolysis of mandelic acid nitrile [532-28-5]
(mp 10℃) with hydrochloric acid. Mandelic acid
nitrile is obtained by the conversion of benzaldehyde [100-52-7] and hydrogen cyanide in the
nascent state (NaCN + HCl) under refrigeration.
Another method of producing mandelic acid is the
hydrolysis of a,a-dichloroacetophenone with
alkali.