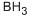Chemical Properties
| Melting point | 2300°C |
| Boiling point | 2550°C |
| Density | 2.34 g/mL at 25 °C (lit.) |
| storage temp. | no restrictions. |
| solubility | H2O: soluble |
| form | Powder |
| color | Brown or dark |
| Specific Gravity | 2.34~2.37 |
| Resistivity | 1.5E12 μΩ-cm, 20 °C |
| Water Solubility | insoluble H2O [MER06] |
| Crystal Structure | Trigonal (rhombohedral) a = 1017 pm α = 65°12' hR105, R3m, β-B type |
| Merck | 13,1333 |
| Exposure limits | ACGIH: TWA 2 mg/m3; STEL 6 mg/m3 |
| Stability | Stable. Substances to be avoided include strong oxidizing agents and strong acids. May decompose on exposure to air - store under nitrogen. Highly flammable. |
| InChIKey | UORVGPXVDQYIDP-UHFFFAOYSA-N |
| CAS DataBase Reference | 7440-42-8(CAS DataBase Reference) |
| NIST Chemistry Reference | Boron(7440-42-8) |
| EPA Substance Registry System | Boron (7440-42-8) |
Safety Information
| Hazard Codes | Xn,F |
| Risk Statements | 22-11-63-62 |
| Safety Statements | 16-24/25-45-36/37/39-27-26 |
| RIDADR | UN 3178 4.1/PG 2 |
| WGK Germany | - |
| RTECS | ED7350000 |
| TSCA | Yes |
| HazardClass | 4.1 |
| PackingGroup | III |
| HS Code | 28045000 |
| Hazardous Substances Data | 7440-42-8(Hazardous Substances Data) |
MSDS
| Provider | Language |
|---|---|
| Boron | English |
| SigmaAldrich | English |
| ACROS | English |
| ALFA | English |
Usage And Synthesis
Boron was discovered by Sir Humphry Davy and J.L. Gay-Lussac in 1808. It is a trivalent non-metallic element that occurs abundantly in the evaporite ores borax and ulexite. Boron is never found as a free element on Earth. Boron appears as charcoal-grey pieces or black powder or as crystalline; is a very hard, black material with a high melting point; and exists in many polymorphs.

Boron has several forms, and the most common one is amorphous boron, a dark powder, non-reactive to oxygen, water, acids, and alkalis. It reacts with metals to form borides. Boron is an essential plant micronutrient. Sodium borate is used in biochemical and chemical laboratories to make buffers. Boric acid is produced mainly from borate minerals by the reaction with sulphuric acid. Boric acid is an important compound used in textile products. The most economically important compound of boron is sodium tetraborate decahydrate or borax, used for insulating fibreglass and sodium perborate bleach. Compounds of boron are used in organic synthesis, in the manufacture of a particular type of glasses, and as wood preservatives. Boron filaments are used for advanced aerospace structures, due to their high strength and light weight.

Boron has several forms, and the most common one is amorphous boron, a dark powder, non-reactive to oxygen, water, acids, and alkalis. It reacts with metals to form borides. Boron is an essential plant micronutrient. Sodium borate is used in biochemical and chemical laboratories to make buffers. Boric acid is produced mainly from borate minerals by the reaction with sulphuric acid. Boric acid is an important compound used in textile products. The most economically important compound of boron is sodium tetraborate decahydrate or borax, used for insulating fibreglass and sodium perborate bleach. Compounds of boron are used in organic synthesis, in the manufacture of a particular type of glasses, and as wood preservatives. Boron filaments are used for advanced aerospace structures, due to their high strength and light weight.
Boron is a yellow or brownish-black powder and may be either crystalline or amorphous. It does not occur free in nature and is found in the minerals borax, colemanite, boronatrocalcite, and boracite. It is insoluble in water but soluble in nitric and sulfuric acids. It is insoluble in cold water, hot water, diethyl ether, and alcohol. If finely divided, it is soluble in most molten metals such as copper, iron, magnesium, aluminum, and calcium. Borates are relatively soluble in water.
Boron is a trivalent, nonmetallic element that occurs abundantly in the evaporite ores, borax and ulexite. Boron is never found as a free element on Earth. Boron as a crystalline is a very hard, black material with a high melting point, and exists in many polymorphs. Boron has several forms, the most common form being amorphous boron, a dark powder, non-reactive to oxygen, water, acids, and alkalis. It reacts with metals to form borides. Boron is an essential plant micronutrient. Sodium borate is used in biochemical and chemical laboratories to make buffers. Boric acid is produced mainly from borate minerals by the reaction with sulfuric acid. Boric acid is an important compound used in textile products. Compounds of boron are used in organic synthesis, in the manufacture of special types of glasses, and as wood preservatives. Boron fi laments are used for advanced aerospace structures owing to their high strength and light weight. It is used as an antiseptic for minor burns or cuts and is sometimes used in dressings. Boric acid was fi rst registered in the United States in 1948 as an insecticide for control of cockroaches, termites, fireants, fleas, silverfish, and many other insects. It acts as a stomach poison affecting the insects' metabolism, and the dry powder is abrasive to the insects' exoskeleton. Boric acid is generally considered to be safe for use in household kitchens to control cockroaches and ants. The important use of metallic boron is as boron fiber. Borate-containing minerals are mined and processed to produce borates for several industrial uses, i.e., glass and ceramics, soaps and detergents, fire retardants and pesticides. The fibers are used to reinforce the fuselage of fi ghter aircraft, e.g., the B-1 bomber. The fibers are produced by vapor deposition of boron on a tungsten fi lament. Pyrex is a brand name for glassware, introduced by Corning Incorporated in 1915. Originally, Pyrex was made from thermal shock-resistant borosilicate glass.
Boron has only three electrons in its outer shell, which makes it more metal than nonmetal.Nonmetals have four or more electrons in their valence shell. Even so, boron is somewhatrelated to metalloids and also to nonmetals in period 2.
It is never found in its free, pure form in nature. Although less reactive than the metalswith fewer electrons in their outer orbits, boron is usually compounded with oxygen andsodium, along with water, and in this compound, it is referred to as borax. It is also found asa hard, brittle, dark-brown substance with a metallic luster, as an amorphous powder, or asshiny-black crystals.
Its melting point is 2,079°C, its boiling point is 2,550°C, and its density is 2.37 g/cm3.
It is never found in its free, pure form in nature. Although less reactive than the metalswith fewer electrons in their outer orbits, boron is usually compounded with oxygen andsodium, along with water, and in this compound, it is referred to as borax. It is also found asa hard, brittle, dark-brown substance with a metallic luster, as an amorphous powder, or asshiny-black crystals.
Its melting point is 2,079°C, its boiling point is 2,550°C, and its density is 2.37 g/cm3.
There are a total of 13 isotopes of boron, two of which are stable. The stableisotope B-10 provides 19.85% of the element’s abundance as found in the Earth’s crust,and the isotope B-11 provides 80.2% of boron’s abundance on Earth.
Boron is the 38th most abundant element on Earth. It makes up about 0.001% of theEarth’s crust, or 10 parts per million, which is about the same abundance as lead. It is notfound as a free element in nature but rather in the mineral borax, which is a compound ofhydrated sodium, hydrogen, and water. Borax is found in salty lakes, dry lake-beds, or alkalisoils. Other naturally occurring compounds are either red crystalline or less dense, dark-brownor black powder.
Boron is also found in kernite, colemanite, and ulexite ores, and is mined in many countries,including the western United States.
Boron is also found in kernite, colemanite, and ulexite ores, and is mined in many countries,including the western United States.
Boron is a semimetal, sometimes classed as a metallic or metalloid or even as a nonmetal.It resembles carbon more closely than aluminum. Although it is extremely hard in its purified form—almost as hard asdiamonds—it is more brittle than diamonds, thus limiting its usefulness. It is an excellentconductor of electricity at high temperatures, but acts as an insulator at lower temperatures.
Boron compounds have been known
for thousands of years, but Boron was not discovered
until 1808 by Sir Humphry Davy and by Gay-Lussac and
Thenard. The element is not found free in nature, but occurs
as orthoboric acid usually in certain volcanic spring waters
and as borates in borax and colemanite. Ulexite, another boron
mineral, is interesting as it is nature’s own version of “fiber
optics.” Important sources of boron are the ores rasorite (kernite)
and tincal (borax ore). Both of these ores are found in the
Mojave Desert. Tincal is the most important source of boron
from the Mojave. Extensive borax deposits are also found in
Turkey. Boron exists naturally as 19.9% 10B isotope and 80.1%
11B isotope. Ten other isotopes of boron are known. High-purity
crystalline boron may be prepared by the vapor phase reduction
of boron trichloride or tribromide with hydrogen on
4-6 The Elements
electrically heated filaments. The impure, or amorphous, boron,
a brownish-black powder, can be obtained by heating the
trioxide with magnesium powder. Boron of 99.9999% purity
has been produced and is available commercially. Elemental
boron has an energy band gap of 1.50 to 1.56 eV, which is higher
than that of either silicon or germanium. It has interesting
optical characteristics, transmitting portions of the infrared,
and is a poor conductor of electricity at room temperature,
but a good conductor at high temperature. Amorphous boron
is used in pyrotechnic flares to provide a distinctive green color,
and in rockets as an igniter. By far the most commercially
important boron compound in terms of dollar sales is Na2B4O7
· 5H2O. This pentahydrate is used in very large quantities in
the manufacture of insulation fiberglass and sodium perborate
bleach. Boric acid is also an important boron compound
with major markets in textile fiberglass and in cellulose insulation
as a flame retardant. Next in order of importance is borax
(Na2B4O7 · 10H2O) which is used principally in laundry products.
Use of borax as a mild antiseptic is minor in terms of dollars
and tons. Boron compounds are also extensively used in
the manufacture of borosilicate glasses. The isotope boron-10
is used as a control for nuclear reactors, as a shield for nuclear
radiation, and in instruments used for detecting neutrons.
Boron nitride has remarkable properties and can be used to
make a material as hard as diamond. The nitride also behaves
like an electrical insulator but conducts heat like a metal. It also
has lubricating properties similar to graphite. The hydrides are
easily oxidized with considerable energy liberation, and have
been studied for use as rocket fuels. Demand is increasing for
boron filaments, a high-strength, lightweight material chiefly
employed for advanced aerospace structures. Boron is similar
to carbon in that it has a capacity to form stable covalently
bonded molecular networks. Carboranes, metalloboranes,
phosphacarboranes, and other families comprise thousands
of compounds. Crystalline boron (99.5%) costs about $6/g.
Amorphous boron (94–96%) costs about $1.50/g. Elemental
boron and the borates are not considered to be toxic, and they
do not require special care in handling. However, some of the
more exotic boron hydrogen compounds are definitely toxic
and do require care.
As early as 1959, boron filaments were introduced as the first of a family of high-strength, high-modulus, low-density reinforcements developed for advanced aerospace applications. A process was engineered by Avco Specialty Materials (Lowell, Massachusetts) and the U.S. Air Force to manufacture boron filaments that had high strength and high stiffness, but low density and, hence, low weight. During the interim, advanced boron fibers have been used as a reinforcement in resin-matrix composites. Boron aluminum has been used for tube-shaped truss members, for reinforcing space vehicle structures, and has also been considered as a fan blade material for turbofan jet engines.These shortcomings led to the development of silicon-carbide (SiC) fibers for some applications.
The principal use of boron filaments is in the form of continuous boronepoxy pre-impregnated tape, commonly known as prepreg. Usually, the resin content is about 30–35% (weight). Boron composites have been used in military aircraft, including helicopters. In addition to aircraft, boron-epoxy composites have been used in tennis, racquetball, squash, and badminton rackets, fishing rods, skis, and golf club shafts, for improving strength and stiffness.
Boron has been used in cutting and grinding tools. Boron is 30–40% harder than silicon carbide and almost twice as hard as tungsten carbide. Boron also has interesting microwave polarization properties. Research (Southern Illinois University) has shown that a single ply of boron epoxy will transmit 98.5% and reflect 0.6% of the incident microwave power when the angle between the grain and the E-field is 90°. This property has been useful in the design of spacecraft antennas and radomes.
The principal use of boron filaments is in the form of continuous boronepoxy pre-impregnated tape, commonly known as prepreg. Usually, the resin content is about 30–35% (weight). Boron composites have been used in military aircraft, including helicopters. In addition to aircraft, boron-epoxy composites have been used in tennis, racquetball, squash, and badminton rackets, fishing rods, skis, and golf club shafts, for improving strength and stiffness.
Boron has been used in cutting and grinding tools. Boron is 30–40% harder than silicon carbide and almost twice as hard as tungsten carbide. Boron also has interesting microwave polarization properties. Research (Southern Illinois University) has shown that a single ply of boron epoxy will transmit 98.5% and reflect 0.6% of the incident microwave power when the angle between the grain and the E-field is 90°. This property has been useful in the design of spacecraft antennas and radomes.
Boron is used to harden metals and as an oxygen scavenger for
copper and other metals. It is used as a reinforcing material for
composites. Boron filaments are used for lightweight but highstrength
building materials for aerospace structures, golf clubs,
and fishing rods. Amorphous boron can produce a green flare,
and is therefore useful in pyrotechnic flares. Boron is also used
in the production of borosilicate glass, which is highly resistant
to thermal shock. An alloy of boron, iron, and neodymium is
used to create a permanent magnet, known as the neodymium
magnet. These magnets are used in magnetic resonance
imaging machines, cell phones, and CD and DVD players.
Boron is also employed as a catalyst in olefin polymerization
and alcohol dehydration. Some boron compounds are used in
the production of insulating fiberglass, bleach, adhesives,
bulletproof vests, and tank armor. The principal consumption
pattern in the United States for boron is for the production of
glass products with minor usage in the production of soaps and
detergents.
Boron has found many uses and has become an important industrial chemical. Boron is used as an alloy metal, and when combined with other metals, it imparts exceptional strength to those metals at high temperatures. It is an excellent neutron absorber used to capture neutrons in nuclear reactors to prevent a runaway fission reaction. As the boron rods are lowered into the reactor, they control the rate of fission by absorbing excess neutrons. Boron is also used as an oxygen absorber in the production of copper and other metals, Boron finds uses in the cosmetics industry (talc powder), in soaps and adhesives, and as an environmentally safe insecticide. A small amount of boron is added as a dope to silicon transistor chips to facilitate or impede the flow of current over the chip. Boron has just three valence electrons; silicon atoms have four. This dearth of one electron in boron s outer shell allows it to act as a positive hole in the silicon chip that can be filled or left vacant, thus acting as a type of switch in transistors. Many of today s electronic devices depend on these types of doped-silicon semiconductors and transistors. Boron is also used to manufacture borosilicate glass and to form enamels that provide a protective coating for steel. It is also used as medication for relief of the symptoms of arthritis. Due to boron s unique structure and chemical properties, there are still more unusual compounds to be explored.
Boron may be prepared by several methods, such as chemical reduction of boron compounds, electrolytic reduction in nonaqueous phase, or by thermal decomposition. Many boron compounds including boron oxides, borates, boron halides, borohydrides, and fluoroborates can be reduced to boron by a reactive metal or hydrogen at high temperatures:
B2O3 + 3Ca → 2B + 3CaO
The metal is obtained as a black amorphous product.
2BCl3 + 3H2 → 2B + 6HCl
High purity grade boron may be prepared by such hydrogen reduction at high temperatures using a hot filament.
Electrolytic reduction and thermal decomposition have not yet been applied in large scale commercial methods. Electrolysis of alkali or alkaline earth borates produces boron in low purity. Electrolytic reduction of fused melts of boron trioxide or potassium tetrafluroborate in potassium chloride yield boron in high purity. Also, boron tribromide or boron hydrides may be thermally dissociated by heating at elevated temperatures.
Impurities from boron may be removed by successive recrystallization or volatilization at high temperatures. Removal of certain impurities such as oxygen, nitrogen, hydrogen or carbon from boron are more difficult and involve more complex steps.
B2O3 + 3Ca → 2B + 3CaO
The metal is obtained as a black amorphous product.
2BCl3 + 3H2 → 2B + 6HCl
High purity grade boron may be prepared by such hydrogen reduction at high temperatures using a hot filament.
Electrolytic reduction and thermal decomposition have not yet been applied in large scale commercial methods. Electrolysis of alkali or alkaline earth borates produces boron in low purity. Electrolytic reduction of fused melts of boron trioxide or potassium tetrafluroborate in potassium chloride yield boron in high purity. Also, boron tribromide or boron hydrides may be thermally dissociated by heating at elevated temperatures.
Impurities from boron may be removed by successive recrystallization or volatilization at high temperatures. Removal of certain impurities such as oxygen, nitrogen, hydrogen or carbon from boron are more difficult and involve more complex steps.
Until the late 1990s elemental boron had not found widespread
use in industry, where cost of production was a major
obstacle. Now, there is increasing use as new applications for
the element are developed in material composites and use in
nanotechnology.
Commercial boron is produced in several ways. (1) Reduction with metals from the abundant B2O3, using lithium, sodium, potassium, magnesium, beryllium, calcium, or aluminum. The reaction is exothermic. Magnesium is the most effective reductant. With magnesium, a brown powder of approximately 90–95% purity is produced. (2) By reduction with compounds, such as calcium carbide or tungsten carbide, or with hydrogen in an electric arc furnace. The starting boron source may be B2O3 or BCl3. (3) Reduction of gaseous compounds with hydrogen. In an atmosphere of a boron halide, metallic filaments or bars at a surface temperature of about 1200 °C will receive depositions of boron upon admission of hydrogen to the process atmosphere. Although the deposition rate is low, boron of high purity can be obtained because careful control over the purity of the starting ingredients is possible. (4) Thermal decomposition of boron compounds, such as the boranes (very poisonous). Boranes in combination with oxygen or H2O are very reactive. In this process, boron halides, boron sulfide, some borides, boron phosphide, sodium borate and potassium borate also can be decomposed thermally. (5) Electrochemical reduction of boron compounds where the smeltings of metallic fluoroborates or metallic borates are electrolytically decomposed. Boron oxide alkali metal oxide–alkali chloride compounds also can be decomposed in this manner.
Elemental boron has found limited use to date in semiconductor applications, although it does possess current-voltage characteristics that make it suitable for use as an electrical switching device. In a limited way, boron also is used as a dopant (p-type) for p?n junctions in silicon. The principal problem deterring the larger use of boron as a semiconductor is the high-lattice defect concentration in the crystals currently available.
Elemental boron has found limited use to date in semiconductor applications, although it does possess current-voltage characteristics that make it suitable for use as an electrical switching device. In a limited way, boron also is used as a dopant (p-type) for p?n junctions in silicon. The principal problem deterring the larger use of boron as a semiconductor is the high-lattice defect concentration in the crystals currently available.
boron: Symbol B. An element ofgroup 13 (formerly IIIB) of the periodictable; a.n. 5; r.a.m. 10.81; r.d.2.34–2.37 (amorphous); m.p. 2300°C;b.p. 2550°C. It forms two allotropes;amorphous boron is a brown powderbut metallic boron is black. Themetallic form is very hard (9.3 onMohs’ scale) and is a poor electrical conductor at room temperature. Atleast three crystalline forms are possible;two are rhombohedral and theother tetragonal. The element isnever found free in nature. It occursas orthoboric acid in volcanic springsin Tuscany, as borates in kernite(Na2B4O7.4H2O), and as colemanite(Ca2B6O11.5H2O) in California. Samplesusually contain isotopes in theratio of 19.78% boron–10 to 80.22%boron–11. Extraction is achieved byvapour-phase reduction of borontrichloride with hydrogen on electricallyheated filaments. Amorphousboron can be obtained by reducingthe trioxide with magnesium powder.Boron when heated reacts withoxygen, halogens, oxidizing acids,and hot alkalis. It is used in semiconductorsand in filaments for specializedaerospace applications.Amorphous boron is used in flares,giving a green coloration. The isotopeboron–10 is used in nuclear reactorcontrol rods and shields. Theelement was discovered in 1808 bySir Humphry Davy and by J. L. Gay-Lussac and L. J. Thenard.
Powdered or fine dust of elemental boron is explosive in air and toxic if inhaled. Several ofthe compounds of boron are very toxic if ingested or if they come in contact with the skin. Thisis particularly true of the boron compounds used for strong insecticides and herbicides.
Boron has been studied extensively for its nutritional importance in animals and humans.
There is a growing body of evidence that boron may be an essential element in animals
and humans. Many nutritionists believe that people would benefi t from more boron and
many popular multivitamins, such as centrum, in the diet. The adverse health effects of
boron on humans is limited. However, ingestion/inhalation causes irritation to the mucous
membrane and boron poisoning.
Short-term exposures to boron in work areas are known to cause irritation of the eye,
the upper respiratory tract, and the naso-pharynx, but the irritation disappears with the
stoppage of further exposure. Ingestion of large amounts of boron (about 30 g of boric acid)over short periods of time is known to affect the stomach, intestines, liver, kidney, and
brain and can eventually lead to death in exposed people.
Boron (B) is a non-metal occupying the first period and
Group 13 (formerly, Ⅲ B) of the Periodic Table. Boron is essential for the growth of new
cells. Its concentration in monocots and dicots varies
between 6 to 18 ppm and 20 to 60 ppm, respectively. In
most crops, the concentration of boron in mature leaf
tissue is over 20 ppm.
Boron is one of the seven micronutrients needed by plants. It exists in soils as a (a) primary rock and mineral, (b) mass combined in soil organic matter or adsorbed on colloidal clay and hydrous oxide surfaces, and (c) borate ion in solution. It occurs as borosilicate to the extent of 20 to 200 ppm in most semi-precious minerals that contain 3 to 4 % boron.
Borosilicate contains varying amounts of iron (Fe), aluminum (Al), manganese (Mn), calcium (Ca), lithium (Li) and sodium (Na). As boron is resistant to weathering, its release from the mineral is slow and, therefore, it cannot meet the need of prolonged and heavy cropping.
Though boron is essential for plants, its requirements and tolerances vary widely from plant to plant. It is required during (a) active cell division, (b) pollen germination, flower formation, fruit and root development, material transportation and cation absorption, (c) new cell development in meristematic tissue, (d) synthesis of amino acids and proteins, (e) nodule formation in legumes, ( f ) translocation of sugars, (g) polymerization of phenolic compounds, and (h) regulation of carbohydrate metabolism. Although boron is required for the growth of agricultural crops, it is not necessary for algae, diatoms, animals, fungi and microorganisms.
Fruits, vegetables, and field crops may suffer from boron deficiency. The first visual symptom is cessation of terminal bud growth, followed by the death of young leaves. Boron deficiency restricts flowering and fruit development, and the symptoms are (a) thickened, wilted or curled leaves, (b) thickened, cracked or water-soaked condition of petioles and stems, and (c) discoloration, cracking or rotting of fruits, tubers or roots. The breakdown of internal root tissues gives rise to darkened areas, referred to as black or brown heart.
The total boron content in soil varies from region to region and soil to soil. In Indian soils, for instance, the total boron content ranges between 4 and 630 mg/kg soil, while the available boron varies from traces to 68 mg/kg soil. Irrigation of arid and semi-arid soils with boron-rich water causes toxicity in plants, which can be reduced with the addition of organic matter.
Boron is available in soils as an organic fraction and is released on decomposition to be partly absorbed by plants and partly lost during leaching. In soil solution, boron is present as a non-ionized molecule (H3BO3) which is absorbed by plant roots and distributed with the transpiration stream. The soil texture, pH and the moisture affect the movement of boron in soils. Coarsetextured sandy soils are low in boron and crops in such soils require additional boron in the form of borax, whereas crops in fine-structured sandy soils do not respond to the added boron. Fine-textured soils retain added boron for longer periods than coarse-textured soils. Clays retain boron more effectively than sands. Plant uptake of boron from clayey soils is larger than that from sandy soils.
The soil pH influences the availability of boron; the higher the pH, the lower the boron uptake and the greater the deficiency. Generally, for the same type of crop, the application rate of a fertilizer containing water-soluble boron is lesser for coarse soils than for fine-textured sandy soils. Apple, alfalfa, asparagus, beet, celery, sunflower are some of the crops requiring high levels of boron (more than 0.5 ppm), whereas carrots, cotton, lettuce, peanuts, peach, sweet potato, tobacco and tomato need only 0.10 to 0.15 ppm of boron. The requirement of barley, beans, citrus, corn, forage grasses, soybeans and strawberry is lower than 0.1 ppm of the available soil boron.
Interaction of boron with nutrients plays a vital role in the efficiency of the use of boron. For instance, boron is particularly effective with phosphorus, potassium and micronutrients, whereas its efficiency suffers with sodium, calcium and magnesium. For a good crop, it is essential to have a correct calcium to boron ratio.
Boron compounds that are used to overcome boron deficiency are borax, boric acid, borosilicate glass or frits, calcium borate (Colemanite) and magnesium borate (Boracite). All boron materials used as fertilizers are stable chemicals and create no storage problem. The various methods by which boron is applied to plants are by drilling, broadcasting and spraying.
The presence of boron in a fertilizer has to be clearly stated on the bag.
Borax (Na2B4O7·10H2O) the most popular boroncontaining fertilizer. For most crops, 15 to 20 kg borax/ha is applied at the time of sowing or transplanting. As boron is readily leached out from the soil and the initial uptake of the plant is large, it is applied as a fused glass to reduce its solubility.
Solubor, a commercial product, is a highly concentrated and completely soluble source of boron (20 %) like borax. It is preferred to borax and is applied as spray or dust directly to the foliage of fruit trees, vegetables and other crops. Colemanite, a naturally occurring calcium borate (Ca2B6O11·5H2O), is less soluble and is also superior to borax.
Boron frits or borosilicate glass containing up to 6 % boron provide boron traces to plants. Borosilicate glass, due to its slow solubility, makes boron available for a longer time than borax. The finely ground form is more effective than the coarse variety.
A dilute solution of boric acid and water is sprayed to be absorbed by the leaves.
Boron is one of the seven micronutrients needed by plants. It exists in soils as a (a) primary rock and mineral, (b) mass combined in soil organic matter or adsorbed on colloidal clay and hydrous oxide surfaces, and (c) borate ion in solution. It occurs as borosilicate to the extent of 20 to 200 ppm in most semi-precious minerals that contain 3 to 4 % boron.
Borosilicate contains varying amounts of iron (Fe), aluminum (Al), manganese (Mn), calcium (Ca), lithium (Li) and sodium (Na). As boron is resistant to weathering, its release from the mineral is slow and, therefore, it cannot meet the need of prolonged and heavy cropping.
Though boron is essential for plants, its requirements and tolerances vary widely from plant to plant. It is required during (a) active cell division, (b) pollen germination, flower formation, fruit and root development, material transportation and cation absorption, (c) new cell development in meristematic tissue, (d) synthesis of amino acids and proteins, (e) nodule formation in legumes, ( f ) translocation of sugars, (g) polymerization of phenolic compounds, and (h) regulation of carbohydrate metabolism. Although boron is required for the growth of agricultural crops, it is not necessary for algae, diatoms, animals, fungi and microorganisms.
Fruits, vegetables, and field crops may suffer from boron deficiency. The first visual symptom is cessation of terminal bud growth, followed by the death of young leaves. Boron deficiency restricts flowering and fruit development, and the symptoms are (a) thickened, wilted or curled leaves, (b) thickened, cracked or water-soaked condition of petioles and stems, and (c) discoloration, cracking or rotting of fruits, tubers or roots. The breakdown of internal root tissues gives rise to darkened areas, referred to as black or brown heart.
The total boron content in soil varies from region to region and soil to soil. In Indian soils, for instance, the total boron content ranges between 4 and 630 mg/kg soil, while the available boron varies from traces to 68 mg/kg soil. Irrigation of arid and semi-arid soils with boron-rich water causes toxicity in plants, which can be reduced with the addition of organic matter.
Boron is available in soils as an organic fraction and is released on decomposition to be partly absorbed by plants and partly lost during leaching. In soil solution, boron is present as a non-ionized molecule (H3BO3) which is absorbed by plant roots and distributed with the transpiration stream. The soil texture, pH and the moisture affect the movement of boron in soils. Coarsetextured sandy soils are low in boron and crops in such soils require additional boron in the form of borax, whereas crops in fine-structured sandy soils do not respond to the added boron. Fine-textured soils retain added boron for longer periods than coarse-textured soils. Clays retain boron more effectively than sands. Plant uptake of boron from clayey soils is larger than that from sandy soils.
The soil pH influences the availability of boron; the higher the pH, the lower the boron uptake and the greater the deficiency. Generally, for the same type of crop, the application rate of a fertilizer containing water-soluble boron is lesser for coarse soils than for fine-textured sandy soils. Apple, alfalfa, asparagus, beet, celery, sunflower are some of the crops requiring high levels of boron (more than 0.5 ppm), whereas carrots, cotton, lettuce, peanuts, peach, sweet potato, tobacco and tomato need only 0.10 to 0.15 ppm of boron. The requirement of barley, beans, citrus, corn, forage grasses, soybeans and strawberry is lower than 0.1 ppm of the available soil boron.
Interaction of boron with nutrients plays a vital role in the efficiency of the use of boron. For instance, boron is particularly effective with phosphorus, potassium and micronutrients, whereas its efficiency suffers with sodium, calcium and magnesium. For a good crop, it is essential to have a correct calcium to boron ratio.
Boron compounds that are used to overcome boron deficiency are borax, boric acid, borosilicate glass or frits, calcium borate (Colemanite) and magnesium borate (Boracite). All boron materials used as fertilizers are stable chemicals and create no storage problem. The various methods by which boron is applied to plants are by drilling, broadcasting and spraying.
The presence of boron in a fertilizer has to be clearly stated on the bag.
Borax (Na2B4O7·10H2O) the most popular boroncontaining fertilizer. For most crops, 15 to 20 kg borax/ha is applied at the time of sowing or transplanting. As boron is readily leached out from the soil and the initial uptake of the plant is large, it is applied as a fused glass to reduce its solubility.
Solubor, a commercial product, is a highly concentrated and completely soluble source of boron (20 %) like borax. It is preferred to borax and is applied as spray or dust directly to the foliage of fruit trees, vegetables and other crops. Colemanite, a naturally occurring calcium borate (Ca2B6O11·5H2O), is less soluble and is also superior to borax.
Boron frits or borosilicate glass containing up to 6 % boron provide boron traces to plants. Borosilicate glass, due to its slow solubility, makes boron available for a longer time than borax. The finely ground form is more effective than the coarse variety.
A dilute solution of boric acid and water is sprayed to be absorbed by the leaves.
Boron (symbol B) is a metallic element closelyresembling silicon. Boron has a specific gravityof 2.31, a melting point of about 2200°C, anda Knoop hardness of 2700 to 3200, equal to aMohs hardness of about 9.3. At 600°C, boronignites and burns with a brilliant green flame.Minute quantities of boron are used in steelsfor case hardening by the nitriding process toform a boron nitride, and in other steels toincrease hardenability, or depth of hardness. Inthese boron steels, as little as 0.003% is beneficial,forming an iron boride, but with largeramounts the steel becomes brittle and susceptibleto hot-short unless it contains titanium orsome other element to stabilize the carbon . Incast iron, boron inhibits graphitization and alsoserves as a deoxidizer. It is added to iron andsteel in the form of ferroboron.
Boron compounds are employed for fluxesand deoxidizing agents in melting metals, andfor making special glasses. Boron, like siliconand carbon, has an immense capacity for formingcompounds, although it has a differentvalence. The boron atom appears to have a lenticularshape, and two boron atoms can make astrong electromagnetic bond, with the boronacting like carbon but with a double ring.
Boron compounds are employed for fluxesand deoxidizing agents in melting metals, andfor making special glasses. Boron, like siliconand carbon, has an immense capacity for formingcompounds, although it has a differentvalence. The boron atom appears to have a lenticularshape, and two boron atoms can make astrong electromagnetic bond, with the boronacting like carbon but with a double ring.
Boron has the atomic number 5 and the symbol B, and is a so-called metalloid. Boron
compounds have been known for many centuries and especially used in the production of glass. At the beginning of the nineteenth century, it was recognised that boron is an essential micronutrient for
plants. A deficiency of boron can lead to deformation in the vegetable growth such as hollow stems and hearts.
Furthermore, the plant growth is reduced and fertility can be affected. In general, boron deficiency leads to
qualitative and quantitative reduction in the production of the crop. Boron is typically available to plants as
boric acid [B(OH)2] or borate [B(OH)4]-. The exact role of boron in plants is not understood, but there is
evidence that it is involved in pectin cross-linking in primary cell walls, which is essential for normal growth
and development of higher plants.
Boron is used in metallurgy as a degasifying
agent and is alloyed with aluminum, iron, and steel
to increase hardness. It is also a neutron absorber in nuclear
reactors. Boron is frequently encountered in a variety of
chemical formulations including boric acid, various borate
salts, borax, and boron soil supplements.
If this chemical gets into the eyes, remove anycontact lenses at once and irrigate immediately for at least15 min, occasionally lifting upper and lower lids. Seek medical attention immediately. If this chemical contacts theskin, remove contaminated clothing and wash immediatelywith soap and water. Seek medical attention immediately. Ifthis chemical has been inhaled, remove from exposure,begin rescue breathing (using universal precautions, including resuscitation mask) if breathing has stopped and CPR ifheart action has stopped. Transfer promptly to a medicalfacility. When this chemical has been swallowed, getmedical attention. Give large quantities of water and inducevomiting. Do not make an unconscious person vomit.
Dust is a flammable solid. Store in a cool, dryplace away from incompatible material, sources of heat andignition. Boron powder may decompose on exposure to airand may have to be stored under a nitrogen blanket.
Boron powder or dust: UN3178 Flammable solid,
inorganic, Hazard Class: 4.1; Labels: 4.1—Flammable solid.
The space lattice of Boron belongs to the tetragonal system with lattice constants a=0.873 nm, c=1.013 nm (c=0.503 nm is also reported). The rhombohedron system is also formed. The rhombohedron is stable near the melting point.
Energy gap: Eg=1.0–1.5 eV
Activation energy : 1.39±0.05 eV
Electron mobility: μe=0.9 cm2 /V s (300 K, 1.8×1016 cm-3 )
Energy gap: Eg=1.0–1.5 eV
Activation energy : 1.39±0.05 eV
Electron mobility: μe=0.9 cm2 /V s (300 K, 1.8×1016 cm-3 )
Boron is ubiquitous in the earth’s crust, and is found in most
soil types in the range 2–100 ppm, and the average concentration
of soil boron is estimated to be 10–20 ppm. The
primary source of boron is the mineral rasorite, also called
kernite. While large areas of the world are boron deficient, high
concentrations are found in parts of western United States, and
throughout China, Brazil, and Russia. The world’s richest
deposits of boron are located in a geographic region that
stretches from the Mediterranean countries inland to
Kazakhstan.
Boron dust may form explosive mixture
in air. Contact with strong oxidizers may cause explosions.
Violent reaction (possible explosion) with concentrated
nitric acid, hydrogen iodide; silver fluoride. Boron is
incompatible with ammonia, bromine tetrafluoride, cesium
carbide, chlorine, fluorine, interhalogens, iodic acid, lead
dioxide, nitric acid, nitrosyl fluoride, nitrous oxide, potassium
nitrite, rubidium carbide. Reacts exothermically with
metals at high temperature above 900° C.
Dispose of contents and container
to an approved waste disposal plant. All federal, state,
and local environmental regulations must be observed.
Elemental boron is non-toxic and common boron compounds, such as borates and boric
acid, have low toxicity (approximately similar to table salt with the lethal dose being
2–3 g/kg) and do not require special precautions while handling. Some of the more
exotic boron hydrogen compounds, however, are toxic as well as highly flammable and do
require special care when handling
Preparation Products And Raw materials
Preparation Products
Boron Supplier
Tel +undefined-86-1523780-4566 +undefined15237804566
Email henankanbeichemical@163.com
Tel +86-024-23769576 +86-15040101888
Email sales@simchoicechem.com
Boron manufacturers
Related articles
Boron: An Element with Exceptional ChemistryApr 7,2024
How many valence electrons does boron have?Mar 12,2024
Is boron nonmetal?Is it good for health?Mar 12,2024
Related Product Information
- Sodium tetraborate
- DI-BORON TRIOXIDE,BORON TRIOXIDE
- Sodium borohydride
- Sodium tetrafluoroborate
- n-Butylboronic acid
1of4
PROMPT×
PROMPT
The What'sApp is temporarily not supported in mainland China
The What'sApp is temporarily not supported in mainland China
Cancel
Determine