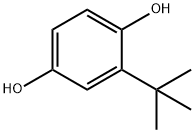
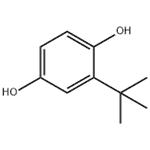
White, crystalline solid having a characteristic odour.

tert-Butylhydroquinone (TBHQ) is an antioxidant used to preserve oils, fats and food items. It is found in vegetable oils and animal fats, varnishes, lacquers, resins, oils field additives, and perfumes.In low concentrations it shows cytoprotective qualities while at higher concentrations it exhibits cytotoxic behavior.
TBHQ was used to study the inactivation of barotolerant strains of Listeria monocytogenes and Escherichia coli.Environment friendly electrode materials for supercapacitors were attained by decorating the surface of graphene nanosheets with TBHQ.
antioxidant in cosmetic products like lipsticks.
TBHQ was used to study the inactivation of barotolerant strains of Listeria monocytogenes and Escherichia coli. Environment friendly electrode materials for supercapacitors were attained by decorating the surface of graphene nanosheets with TBHQ. TBHQ is frequently used as a food preservative. Inhibition of oxidative rancidity in frozen cooked fish flakes by tert-butylhydroquinone. Nuclear factor E2-related factor 2-dependent antioxidant response element activation is by tert-butylhydroquinoneoccurring preferentially in Astrocytes conditions.
tert-Butylhydroquinone (TBHQ) is an antioxidant that exhibits an excellent stabilizing effect in unsaturated fats and oils. It has good solubility in fats and oils, with a maximum usage level of 0.02% based on the weight of the fat or oil or the fat content of the food product. It shows no discoloration in the presence of iron and produces no discernible flavor or odor. It can be combined with BHA and BHT. It is used in edible fats and vegetable oils to retard rancidity. It is used in potato chips and dry cereal. It is also termed butylhydroquinone and mono-tertiary-butylhydroquinone.
ChEBI: A member of the class of hydroquinones in which one of the ring hydrogens of hydroquinone is replaced by a tert-butyl group.
White to light tan crystalline powder or a fine beige powder. Very slight aromatic odor.
Phenols, such as tert-Butylhydroquinone, do not behave as organic alcohols, as one might guess from the presence of a hydroxyl (-OH) group in their structure. Instead, they react as weak organic acids. Phenols and cresols are much weaker as acids than common carboxylic acids (phenol has Ka = 1.3 x 10^[-10]). These materials are incompatible with strong reducing substances such as hydrides, nitrides, alkali metals, and sulfides. Flammable gas (H2) is often generated, and the heat of the reaction may ignite the gas. Heat is also generated by the acid-base reaction between phenols and bases. Such heating may initiate polymerization of the organic compound. Phenols are sulfonated very readily (for example, by concentrated sulfuric acid at room temperature). The reactions generate heat. Phenols are also nitrated very rapidly, even by dilute nitric acid. Nitrated phenols often explode when heated. Many of them form metal salts that tend toward detonation by rather mild shock. tert-Butylhydroquinone is incompatible with oxidizers.
tert-Butylhydroquinone is combustible.
Flammability and Explosibility
Non flammable
This antioxidant has seldom been reported as a sensitizer,
mainly in cosmetics (lipsticks, lip-gloss, hair
dyes) or in cutting oils. Simultaneous/cross-reactions
have been described to butylhydroxyanisole (BHA)
and less frequently to butylhydroxytoluene (BHT), but
not to hydroquinone
Recrystallise the hydroquinone from H2O or MeOH and dry it in a vacuum at 70o. Store it in a dark container. [Stroh et al. Angew Chem 69 699 1957, Beilstein 6 IV 6013.]