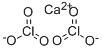Calcium chlorate forms white to yellow deliquescent crystals. Molecular weight=243; Specific gravity(H2O:1)=2.71; Freezing/Melting point=340°(loses H2Oof crystallization at .100°). Hazard Identification (basedon NFPA-704 M Rating System): Health 1, Flammability 0,Reactivity 0; oxidizer. Highly soluble in water.
Calcium chlorate forms white to yellow deliquescent crystals.
Calcium chlorate Ca(ClO3)2 is the chemical compound
formed from calcium and the chlorate anion. Like KClO3,
it is a strong oxidizer and can be used in pyrotechnic
formulations. Its molecular weight is 206.98 g/mol. Its solubility in water is 209 g/100 ml at 20°C. It is slightly
soluble in alcohol. Calcium chlorate has a melting point
of 325°C and its density is 2.71 g/cm3. Its CAS number
is 10017-74-3. A dihydrate, Ca(ClO3)2·2H2O, forms as
white monoclinic crystals, which are decomposed by
heating above 150°C. No structural data is available.
Calcium chlorate has been used as an herbicide, like sodium chlorate. It is occasionally used in photography, dusting powder to kill poison ivy, pyrotechnics, as an oxidizer and pink flame colorant. Its hygroscopic nature and incompatibility with other common pyrotechnic materials (such as sulfur) limit its utility in these applications.
Calcium chlorate can be prepared by the Liebig
process which is also used to prepare the alkali chlorates.
In Liebig’s process, chlorine is passed into milk of lime, at or above a temperature of 1000°C, the
apparent reaction being:
6Ca(OH)2+ 6Cl2→5CaCl2+ Ca(CIO3)2+ 6H2O.
But this may comprise two minor reactions:
2Ca (OH)2+ 2Cl2→CaCl2+ Ca(OCl)2+ 2H2O;
3Ca(OCI)2→2CaCl2+ Ca(CIO3)2
This salt can also be prepared from calcium chloride
by an electrochemical method:
CaCl2 (aq)+ 6H2O (aq)+ e-→Ca(ClO3)2+ 6H2
using a platinum anode and a rotating stainless steel
cathode. Yields of the dihydrate up to 78% have been
achieved. The hydrogen produced can be “burned” to reform water.
Calcium chlorate appears as a white crystalline solid. It forms a very flammable mixture with combustible materials and this mixture may be explosive if the combustible material is finely divided. The mixture can be ignited by friction. Contact with strong sulfuric acid can cause fires or explosions. When mixed with ammonium salts, spontaneous decomposition and ignition may result. Prolonged exposure of the material to fire or heat can result in an explosion. Calcium chlorate is used in photography, in pyrotechnics, and as a herbicide.
An oxidizing agent. Liberates explosive chlorine dioxide gas in the presence of strong acid. Heating a moist mixture with a dibasic organic acid liberates chlorine dioxide and carbon dioxide. Mixtures with ammonium salts, powdered metals, silicon, sulfur, or sulfides are readily ignited and potentially explosive [Bretherick 1979 p. 806]. A combination with finely divided aluminum can explode by heat, percussion, or friction [Mellor 2:310 1946-47].
Oxidizer, dangerous fire risk, forms explo-
sive mixtures with combustible materials.
Inhalation of dust causes irritation of upper respiratory system. Dust irritates eyes and skin. Ingestion causes abdominal pain, nausea, vomiting, diarrhea, pallor, shortness of breath, unconsciousness.
Behavior in Fire: When involved in a fire, may cause an explosion. Irritating gases may be generated when heated.
Moderately toxic by
ingestion and intraperitoneal routes. A
powerful oxidant. Incompatible with Al, As,
C, Cu, charcoal, MnO2, metal sulfides, S,
dibasic organic acids, organic matter, P.
When heated to decomposition it emits
toxic fumes of Cl-. See also CHLORATES
for fire, disaster, and explosion hazards.
Calcium chlorate is used in making
fireworks, herbicides (weed killers) and in photography.
Incompatibilities: A strong oxidizer. Reacts, possibly with
risk of fire and explosion, with acids (especially organic
acids), reducing agents; aluminum, arsenic, chemically
active metals; combustible materials; ammonium compounds; charcoal, copper, cyanides; manganese dioxide,
metal sulfides; phosphorus, sulfur
If this chemical gets into the eyes, remove anycontact lenses at once and irrigate immediately for at least15 min, occasionally lifting upper and lower lids. Seekmedical attention immediately. If this chemical contacts theskin, remove contaminated clothing and wash immediatelywith soap and water. Seek medical attention immediately. Ifthis chemical has been inhaled, remove from exposure,begin rescue breathing (using universal precautions, including resuscitation mask) if breathing has stopped and CPR ifheart action has stopped. Transfer promptly to a medicalfacility. When this chemical has been swallowed, getmedical attention. Give large quantities of water and inducevomiting. Do not make an unconscious person vomit.Note to physician: Treat for methemoglobinemia.Spectrophotometry may be required for precise determination of levels of methemoglobin in urine.
Color Code—Yellow: Reactive Hazard; Store in alocation separate from other materials, especially flammablesand combustibles. Store in tightly closed containers in a cool,well-ventilated area away from strong acids (such ashydrochloric, sulfuric, and nitric) and chemically active metals(such as potassium, sodium, magnesium and zinc). Calciumchlorate must be stored to avoid contact with organic matter,ammonium compounds, aluminum, copper, cyanides, flammable vapors, and other oxidizable materials since violent reactions occur. Avoid storage on wood floors. Friction, heat, orphysical shocks may cause calcium chlorate to ignite andexplode. Wherever calcium chlorate is used, handled, manufactured, or stored, use explosion-proof electrical equipmentand fittings. See OSHA Standard 1910.104 and NFPA 43ACode for the Storage of Liquid and Solid Oxidizers for detailedhandling and storage regulations.
UN1452 Calcium chlorate, Hazard Class: 5.1;
Labels: 5.1-Oxidizer. UN2429 Calcium chlorate, aqueous
solution, Hazard Class: 5.1; Labels: 5.1-Oxidizer
Calcium chlorate is used in making
fireworks, herbicides (weed killers) and in photography.
Incompatibilities: A strong oxidizer. Reacts, possibly with
risk of fire and explosion, with acids (especially organic
acids), reducing agents; aluminum, arsenic, chemically
active metals; combustible materials; ammonium compounds; charcoal, copper, cyanides; manganese dioxide,
metal sulfides; phosphorus, sulfur
For barium chlorate, the
UN recommends using a vast volume of a reducing
agent (bisulfites, ferrous salts or hypo) followed by neutralization and flushing to the sewer with abundant water. This
should be applicable here as well.