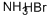Ammonium bromide is soluble in water and alcohol and is not particularly hazardous under normal conditions.
Colorless rhombic crystal; odor of ammonia; sublimes at 60°C; very soluble in cold water; decomposes in hot water; slightly soluble in alcohol; insoluble in acetone.
Colorless crystals made by hydrobromic acid acting upon
ammonium hydroxide followed by crystallization. Soluble in
ether, alcohol, and water, ammonium bromide was used as
a halide in collodion formulas and as a restrainer in alkaline
developers.
manufacture of photographic films, plates, and papers; in process engraving and lithography; fireproofing of wood; in corrosion inhibitors.
A chemical used for its bromide ion
ChEBI: An ammonium salt composed of ammonium and bromide ions in a 1:1 ratio.
White odorless crystals or granules that become yellow upon exposure to air. Sinks and mixes in water.
Bromine trifluoride reacts explosively with the following Ammonium bromide, ammonium chloride, ammonium iodide [Mellor 2 Supp. 1:165 1956].
INHALATION: Dust irritating - disturbed behavior, sedation. EYES: Slight irritation. SKIN: Slight irritation only with repeated or prolonged contact. INGESTION: Weakness, nervousness, anorexia, confusion, hallucinations, drowsiness, irritability, ataxia, vertigo, skin rash.
Special Hazards of Combustion Products: Material decomposes into N 2 and HBr or Br 2 under extreme temperatures.
Flammability and Explosibility
Non flammable
It crystallises from 95% EtOH and is slightly hygroscopic.