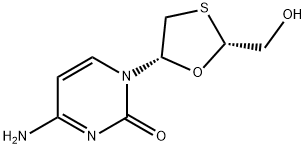
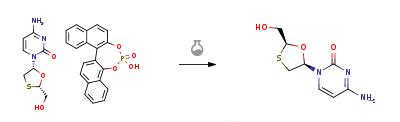
[1] ROUSSOSA. Lamivudine treatment for acute severe hepatitis B: report of a case and review of the literature.[J]. Acta gastro-enterologica Belgica, 2008, 71 1: 30-32.
[2] ARTSE J WainbergM A. Mechanisms of nucleoside analog antiviral activity and resistance during human immunodeficiency virus reverse transcription.[J]. Antimicrobial Agents and Chemotherapy, 1996, 40 3: 527-540. DOI:
10.1128/AAC.40.3.527.
[3] COATESJ A. (-)-2’-deoxy-3’-thiacytidine is a potent, highly selective inhibitor of human immunodeficiency virus type 1 and type 2 replication in vitro.[J]. Antimicrobial Agents and Chemotherapy, 1992, 36 4: 733-739. DOI:
10.1128/AAC.36.4.733.
[4] KONGHUI. Targeted P2X7/NLRP3 signaling pathway against inflammation, apoptosis, and pyroptosis of retinal endothelial cells in diabetic retinopathy.[J]. Cell Death & Disease, 2022, 13 4: 336. DOI:
10.1038/s41419-022-04786-w.
[5] KUMAR P N, PATEL P. Lamivudine for the treatment of HIV[J]. Expert Opinion on Drug Metabolism & Toxicology, 2010, 6: 105-114. DOI:
10.1517/17425250903490418.
[6] PERRYC M FauldsD. Lamivudine. A review of its antiviral activity, pharmacokinetic properties and therapeutic efficacy in the management of HIV infection.[J]. Drugs, 1997, 53 4: 657-680. DOI:
10.2165/00003495-199753040-00008.
[7] RAJURKARMIHIR. Reverse Transcriptase Inhibition Disrupts Repeat Element Life Cycle in Colorectal Cancer.[J]. Cancer discovery, 2022, 12 6: 1462-1481. DOI:
10.1158/2159-8290.CD-21-1117.