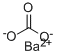Industrial product is white powder. Almost insoluble in water, soluble in alcohol, soluble in acid and ammonium chloride solution.
(1) Barium carbonate is mainly used for the manufacture of optical glass, funnels, and barium magnetic materials, the manufacture of other barium salts, ceramic, enamel, paint, welding rod feed
(2) It is important chemical raw materials for the production of funnels, magnetic materials and advanced optical glass.
(3) It is used for fireworks, flares preparation, also used for auxiliary material of ceramic coatings and optical glass.
(4) It is used for the manufacture of electronic ceramics, PTC thermistors, capacitors and other electronic components.
(5) Barium carbonate is mainly used for the manufacture of optical glass, CRT glass, and barium magnetic materials and capacitors, it is also used for carburizing steel and metal surface treatment. It is the raw material of manufacturing other barium salts and ceramic, enamel, pigments, paints, rubber, electrode. It is also used as rodenticides and purifying agent, oxidation catalyst.
(6) It is mainly used for the manufacture of optical glass, CRT glass, and barium magnetic materials and capacitors, it is also used for carburizing steel and metal surface treatment. It is the raw material of manufacturing other locks salt and ceramic, enamel, pigments, paints, rubber, electrode. It is also used as purifying agent and rodenticides.
(7) It is used for the production of funnels, electronic ceramics and purified water, producing pigment, paint or other barium salts, it is used for the manufacture of optical glass, barium magnetic materials, it is important chemical raw material of manufacturing funnels, magnetic materials and advanced optical glass. It is used for fireworks, flares preparation, it is also used as auxiliary material of ceramic coatings and optical glass, it is used for manufacturing electronic ceramics, PTC thermistor, capacitor of electronic components, it is mainly used in the manufacture of optical glass, CRT glass and barium magnetic materials and capacitors, it is also used for carburizing steel and metal surface treatment. It is raw material of manufacturing other barium salts and ceramic, enamel, pigments, paints, rubber, electrode materials. It is also used as rodenticides and purifying agent, oxidation catalyst. It is mainly used in the manufacture of optical glass, CRT glass and barium magnetic.
Producing piezoelectric ceramic capacitors
Barium carbonate is widely used in high-voltage AC capacitors, ultrasonic transducer devices and piezoelectric ceramic capacitor manufacturing.
The piezoelectric effect was first discovered on quartz crystal by the Curie brothers in 1880, the crystal which can produce the piezoelectric effect was called piezoelectric crystal. The class of piezoelectric crystal is single crystal, such as quartz (SiO2), sodium potassium tartrate (it is also known Luose salt, NaKC4H4O6 * H2O), BGO (Bi12GeO20) and the like. Another class of piezoelectric crystals is called piezoelectric ceramics such as barium titanate (BaTiO3), lead zirconate titanate [Pb (ZrxTirx) O3, code PZT], magnesium niobate lead zirconate titanate of Japan made [Pb (Mg1/3Nb2/3) O3 added PZT, code PCM], manganese, antimony, lead zirconate titanate of China made [Pb (Mn1/2Sb2/3) O3 added PIT code PMS] and so on.
Piezoelectric ceramic materials have three major categories:
PZT materials (PbZrTiO3): titania, zirconia, lead oxide, strontium oxide, niobium oxide, lanthanum oxide and the like.
PMN class materials (PbNbMgO3): niobium oxide, magnesium oxide, lead oxide, strontium oxide, lanthanum oxide and the like.
Barium titanate type materials (BaTiO3): titanium dioxide, barium carbonate, strontium carbonate.
The above information is edited by ChemicalBook editor Maggie.
This product is toxic virulent. Chronic poisoning mainly accumulates in the bone, causing bone marrow leukosis. Severe acute poisoning can cause acute gastrointestinal disorders, tendon reflexes, convulsions, muscle paralysis. Emergency treatments include gastric lavage, enema, taking measures like vomiting. Maximum allowable concentration is 0.5 mg/m3. Production should wear overalls, masks, gloves.
Toxicity: See barium carbonate.
(1) Carbonation method: carbon dioxide is passed through barium sulfide solution (see preparation of barium sulfide) to carbonize, the obtained barium carbonate slurry is desulfurization washed, vacuum filtration, dried at 300℃ and pulverized to obtain barium carbonate products. its
BaS + CO2 + H2O→ BaCO3↓+ H2S↑
Metathesis method: barium sulfide and ammonium carbonate proceeds metathesis reaction, and then washing, filtering and drying to obtain bismuth products. its
BaS + (NH4) 2CO3 → BaCO3 ↓ + (NH4) 2S
Poison nepheline conversion method: witherite ore reacts with ammonium salt to generate soluble barium salt, and ammonium carbonate is recycled and re-used, ammonium carbonate is added into soluble barium salt to precipitate refined barium carbonate, it is filtered and dried to obtain barium carbonate products. The obtained mother liquor is recycled use.
(2) Carbonization method. Reaction equation:
BaS + CO2 → BaCO3 ↓ + H2S ↑
Operational method: 1L barium sulfide aqueous solution of 180g/L reacts with carbon dioxide at 70℃ to produce barium carbonate precipitation. Precipitation is mixed with 10ml 25% aqueous ammonia, at 60℃ desulfurization for 20min. Filtered, and dried to obtain the precipitated barium carbonate, product contains 0.46% sulfur.
Metathesis method. Reaction equation:
BaCl2 + NH4HCO3 + NH4OH → BaCO3 ↓ + 2NH4Cl + H2O
Operational method: At 30℃, 23.6L containing ammonium bicarbonate 56.1g /L aqueous solution and 1.36L ammonia is mixed in 50L kettle and then at 50℃, within 2h dropping 21.6L 200.3g/L barium chloride solution, it is stirred for 30min at 40℃. Filtered, dried at 140 ℃, pulverized to obtain barium carbonate which average particle diameter d is 0.8μm, purity is 99.8%.
Witherite method. Reaction equation:
BaCO3 + 2NH4Cl → BaCl2 + 2NH3 + H2O + CO2
BaCL2 + (NH4) 2CO3 → BaCO3 ↓ + 2NH4Cl
Operational method: 100 g 80% of toxic heavy powder (120 mesh) and 60g ammonium chloride is well mixed, it is rapidly put into 650℃ muffle furnace and reacts for 3min. After cooling, it is flooded, filtered, removed debris to obtain barium chloride solution, 36g ammonium carbonate is added, precipitating barium carbonate, it is filtered, dried and pulverized, the conversion rate is 91%.
(3) Operational method: Dry granulation method: heavy precipitation barium carbonate is sieved, it is placed within the raw materials warehouse and stirred well, mixed, degassed, the material goes through the rotary feeder, lying vertical screw conveyor rollers press rollers compressed tablets, producer thickness is 3.7~4 mm. The material rolled into sheet is input granulator, by adjusting its speed and aperture size sieve to adjust the particle size distribution of semi-finished products. Semi-finished products use knife pulse pneumatic conveying method which semi-finished products input the vibration feeder to sieve, particles larger than 20 mesh is back to granulator, particle 65 mesh or less is back to raw materials warehouse, particle between 65 mesh to 20 mesh is transported to packaging warehouse for finished packaging to obtain particles of barium carbonate.
Wet granulation of barium carbonate: Precipitation system is filtered and separated to obtain cake of containing water in manufacturing process system, and the filter cake should control the water content to about 20%. Under the action of the rotating blades, the material is rapidly mixed well, and kneaded, mediated to form dense particles, the wet pellets is put into the rotary kiln direct fire, sintered at 800~1200℃. After whole particles, screening, iron removal, weighing and packing to obtain particles of barium carbonate.
Hazards & Safety Information
Category: Pesticide.
Toxicity grading: Highly toxic.
Acute oral toxicity-rat LD50: 418 mg/kg; Oral-Mouse LD50: 200 mg/kg
Heating flammability hazard characteristics: when be heated, it can produce toxic barium oxide fumes; eating it can cause gastric ulcer, muscle weakness, paresthesia, numbness, diarrhea, vomiting.
Storage characteristics: Treasury ventilation low-temperature drying; it should be stored separately with acids, food additives.
Extinguishing agent: Water, carbon dioxide, dry, sandy soil.
Professional standards: TWA 0.5 mg (barium)/cubic meter; STEL 1.5 mg (barium)/cubic meter.
Barium carbonate has the molecular formula of
BaCO3 and the molecular weight of 197.3359 g/mol. Its
CAS number is 513-77-9. Barium carbonate has only
one stable form (aragonite-type structure) and temperature
of precipitation has no effect on crystal form, unlike
that of calcium or magnesium carbonates.
Barium carbonate, BaCO3, also known as witherite, is a white powder that is soluble in acids,with the exception of sulfuric acid.It has a melting point of 174°C and is used in television picture tubes, rodenticide, optical glass and ceramic flux.

Barium oxide, BaO, is manufactured by decomposition of barium carbonate.
White powder; orthorhombic crystal system; density 4.286 g/cm3; refractive index 1.60; hardness 3.50 Mohs; melts at 811°C; insoluble in water (c. 25 mg/L at 25°C); Ksp 2.0 x 10-9.
Barium carbonate is found in nature as mineral witherite. The compound has many major commercial applications in brick, glass, ceramics, oil-drilling, photographic and chemical industries. It is mixed with wet clay to immobilize many water-soluble salts in making uniform red bricks. In the glass industry, barium is added to glass as barium carbonate or barium oxide to improve the refractive index of optical glass; also to promote sintering and lower the viscosity of melted glass to make glass bead formation easy. It is used in the manufacture of television picture tubes and photographic paper. Another important application involves its use as a fluxing ingredient in ceramic industry for enamels, glazes and ceramic bodies. Barium carbonate is used in oil-well drilling to insolubilize gypsum and inhibit coagulation; in ferrous metallurgy for steel carburizing; in chlor-alkali cells for treating salt brines to remove sulfates; and to make ferrite, and barium titanate. Many barium salts are prepared from barium carbonate.
Rat poison; in ceramics, paints, enamels, marble substitutes, rubber; manufacture of paper, barium salts, electrodes, optical glasses; as an analytical reagent.
Barium carbonate has many major commercial applications
in brick, glass, ceramics, oil-drilling, photographic
and chemical industries. It is mixed with wet clay to
immobilize many water-soluble salts in making uniform
red bricks. In the glass industry, barium is added to glass
as barium carbonate or barium oxide to improve the
refractive index of optical glass and also to promote sintering
and lower the viscosity of melted glass to make
glass bead formation easy. It is used in the manufacture
of television picture tubes and photographic paper.
Another important application involves its use as a fluxing
ingredient in ceramic industry for enamels, glazes
and ceramic bodies. Barium carbonate is used in oil
well drilling to insolubilize gypsum and inhibit coagulation;
in ferrous metallurgy for steel carburizing; in chloralkali
cells for treating salt brines to remove sulfates; and
to make ferrites, and barium titanate. Many barium salts
are prepared from barium carbonate.
Preparation of barium standard solution. It is used in ceramic glazes, cement, bricks and in rat poison.
Barium
carbonate is made commercially from barium sulfide
either by treatment with sodium carbonate or ammonium
carbonate at 60 to 70°C or by passing CO2 gas
through a soluble Ba2+
solution at 40 to 90°C.
witherite is a mineral form of bariumcarbonate, BaCO3.
barium carbonate: A white insolublecompound, BaCO3; r.d. 4.43. It decomposeson heating to give bariumoxide and carbon dioxide:
BaCO3(s) → BaO(s) + CO2(g)
The compound occurs naturally asthe mineral witherite and can be preparedby adding an alkaline solutionof a carbonate to a solution of a bariumsalt. It is used as a raw materialfor making other barium salts, as aflux for ceramics, and as a raw materialin the manufacture of certaintypes of optical glass.
Barium carbonate is a white powder. Barium carbonate is insoluble in water and soluble in most acids, with the exception of sulfuric acid. Barium carbonate has a specific gravity of 4.275. Barium carbonate is toxic by ingestion.
Barium carbonate is insoluble in water and soluble in most acids, with the exception of sulfuric acid.
Salts, basic, such as Barium carbonate, are generally soluble in water. The resulting solutions contain moderate concentrations of hydroxide ions and have pH's greater than 7.0. They react as bases to neutralize acids. These neutralizations generate heat, but less or far less than is generated by neutralization of the bases in reactivity group 10 (Bases) and the neutralization of amines. They usually do not react as either oxidizing agents or reducing agents but such behavior is not impossible.
(INGESTION ONLY): excessive salivation, vomiting, severe abdominal pain, and violent purging with watery and bloody stools; a slow and often irregular pulse and a transient elevation in arterial blood pressure; tinnitus, giddiness and vertigo; muscle twitchings, progressing to convulsions and/or paralysis; dilated pupils with impaired accommodation; confusion and increasing somnolence, without coma; collapse and death from respiratory failure and cardiac arrest.
Non-combustible, substance itself does not burn but may decompose upon heating to produce corrosive and/or toxic fumes. Some are oxidizers and may ignite combustibles (wood, paper, oil, clothing, etc.). Contact with metals may evolve flammable hydrogen gas. Containers may explode when heated.
Poison by ingestion,
intravenous, and intraperitoneal routes.
Human systemic effects by ingestion:
stomach ulcers, muscle weakness,
paresthesias and paralysis, hypermotility,
diarrhea, nausea or vomiting, lung changes.
Experimental reproductive effects.
Incompatible with BrF3 and 2-
furanpercarboxylic acid. See also BARIUM
COMPOUNDS (soluble).