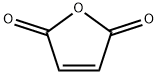
Maleic Anhydride (MAN) is an organic compound with the chemical formula C4H203. It is the acid anhydride of maleic acid and in its pure state it is a colourless or white solid with an acrid odour. Maleic anhydride is truly a remarkable molecule in that it possesses two types of chemical functionality making it uniquely useful in chemical synthesis and applications.

Maleic Anhydride is a multifunctional chemical intermediate with applications in several fields of chemical industry. Its major end use is as feedstock in the production of unsaturated polyester resins (UPR).
These resins are used both in glass-reinforced and in unreinforced applications. The UPR end uses includes a wide range of applications in construction, marine and automobile industries.
In addition, Maleic Anhydride can also be used as raw material in the production of 1,4-butanediol (BDO), gamma- butyrolactone and tetrahydrofuran (THF). It is important to highlight that BDO is one of the world's fastest growing chemicals in the last years.
Maleic anhydride is colorless needles, white lumps, or pellets. Irritating, choking odor. Dissolves in water to produce maleic acid. Dissolves in ethanol and produces esters.
White, hydroscopic crystals (usually shipped as briquettes). Odor threshold concentration is 0.32
ppm (quoted, Amoore and Hautala, 1983).
In the manufacture of polyester resins,
fumaric acid, agricultural pesticides, and alkyl
resins
Maleic Anhydride is heterocyclic compound used in the manufacture of unsaturated polyester resins. Maleic Anhydride has a wide range of other applications; it is used in synthetic tensides, insecticides, herbicides and fungicides.
In Diels-Alder syntheses (as a dienophile), manufacture of alkyd-type of resins, dye intermediates, pharmaceuticals, agricultural chemicals (maleic hydrazide, malathion), in copolymerization reactions.
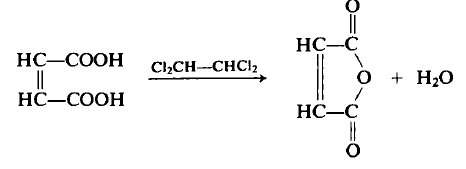
To a flask equipped with a Dean-Stark trap, condenser, and mechanical stirrer is added 116 gm (1.0 mole) of maleic acid and 120 ml of tetrachloroethane. The contents are heated, the water (18 ml, 1.0 mole) distilled off as the azeotrope, and the residue distilled under reduced pressure to afford 87.7 gm (89.5%) of the anhydride, b.p. 82-84°C (15 mm), m.p. 53°C. The residue remaining in the flask consists of about 10 gm of fumaric acid, m.p. 287°C.
Fumaric and maleic acids both give maleic anhydride on heating. Fumaric acid must first be heated to a higher temperature to effect its conversion to maleic acid prior to its dehydration.
ChEBI: Maleic anhydride is a cyclic dicarboxylic anhydride that is the cyclic anhydride of maleic acid. It has a role as an allergen. It is a cyclic dicarboxylic anhydride and a member of furans.
Maleic anhydride was traditionally manufactured by the oxidation of benzene or other aromatic compounds. As of 2006, only a few smaller plants continue to use benzene; due to rising benzene prices, most maleic anhydride plants now use n-butane as a feedstock.
In both cases, benzene and butane are fed into a stream of hot air, and the mixture is passed through a catalyst bed at high temperature. The ratio of air to hydrocarbon is controlled to prevent the mixture from catching on fire. Vanadium pentoxide and molybdenum trioxide are the catalysts used for the benzene route, whereas vanadium and phosphorus oxides are used for the butane route.
2 CH3CH2CH2CH3 + 7 O2 → 2 C2H2(CO)2O + 8 H2O.
The chemistry of maleic anhydride is very rich, reflecting its ready availability and bifunctional reactivity. It hydrolyzes, producing maleic acid, cis-HOOC–CH=CH–COOH. With alcohols, the halfester is generated, e.g., cis-HOOC–CH=CH–COOCH3.
Maleic anhydride is a potent dienophile in Diels-Alder reactions. It is also a ligand for low-valent metal complexes, examples being Pt(PPh3)2(MA) and Fe(CO)4(MA).
Maleic anhydride dimerizes in a photochemical reaction to form cyclo butane tetra carboxylic dianhydride (CBTA). This compound is used in the production of polyimides and as an alignment film for liquid crystal displays.
Colorless crystalline needles, flakes, pellets, rods, briquettes, lumps or a fused mass. Melts at 113°F. Shipped both as a solid and in the molten state. Vapors, fumes and dusts strong irritate the eyes, skin and mucous membranes. Flash point 218°F. Autoignition temperature 890°F. Used to make paints and plastics and other chemicals.
Soluble in water. Reacts slowly with water to form maleic acid and heat.
Maleic anhydride react vigorously on contact with oxidizing materials. Reacts exothermically with water or steam. Undergoes violent exothermic decomposition reactions, producing carbon dioxide, in the presence of strong bases (sodium hydroxide, potassium hydroxide, calcium hydroxide), alkali metals (lithium, sodium, potassium), aliphatic amines (dimethylamine, trimethylamine), aromatic amines (pyridine, quinoline) at temperatures above 150° C [Vogler, C. A. et al., J. Chem. Eng. Data, 1963, 8, p. 620]. A 0.1% solution of pyridine (or other tertiary amine) in Maleic anhydride at 185°C gives an exothermic decomposition with rapid evolution of gas [Chem Eng. News 42(8); 41 1964]. Maleic anhydride is known as an excellent dienophile in the Diels-Alder reaction to produce phthalate ester derivatives. These reactions can be extremely violent, as in the case of 1-methylsilacyclopentadiene [J. Organomet., Chem., 1979, 179, c19]. Maleic anhydride undergoes a potentially explosive exothermic Diels-Alder reaction with 1-methylsilacyclopenta-2,4-diene at 150C [Barton, T. J., J. Organomet. Chem., 1979, 179, C19], and is considered an excellent dieneophile for Diels-alder reactions [Felthouse, Timothy R. et al. "Maleic anhydride , Maleic Acid, and Fumaric Acid." Kirk-Othmer Encyclopedia of Chemical Technology. John Wiley & Sons, Inc. 2005].
Irritant to tissue. Dermal and respiratory
sensitization. Questionable carcinogen.
Inhalation causes coughing, sneezing, throat irritation. Skin contact causes irritation and redness. Vapors cause severe eye irritation; photophobia and double vision may occur.
Behavior in Fire: When heated above 300°F in the presence of various materials may generate heat and carbon dioxide. Will explode if confined.
Flammability and Explosibility
Non flammable
Poison by ingestion and
intraperitoneal routes. Moderately toxic by
skin contact. A corrosive irritant to eyes,
skin, and mucous membranes. Can cause
pulmonary edema. Questionable carcinogen
with experimental tumorigenic data.
Mutation data reported. A pesticide.
Combustible when exposed to heat or
flame; can react vigorously on contact with
oxidizing materials. Explosive in the form of
vapor when exposed to heat or flame.
Reacts with water or steam to produce heat.
Violent reaction with bases (e.g., sodmm
hydroxide, potassium hydroxide, calcium
hydroxide), dkah metals (e.g., sodium,
potassium), amines (e.g., dimethylamine,
triethylamine), lithium, pyridine. To fight
fire, use alcohol foam. Incompatible with
cations. When heated to decomposition
(above 150℃) it emits acrid smoke and
irritating fumes. See also ANHYDRIDES.
Maleic anhydride is used in unsaturated polyester resins; Agricultural chemical, and lubricating additives; in the manufacture of unsaturated polyester
resins; in the manufacture of fumaric acid; in alkyd resin manufacture; in the manufacture of pesticides e.g., malathion, maleic hydrazide, and captan).
If this chemical gets into the eyes, remove anycontact lenses at once and irrigate immediately for at least15 min, occasionally lifting upper and lower lids. Seek medical attention immediately. If this chemical contacts theskin, remove contaminated clothing and wash immediatelywith soap and water. Seek medical attention immediately. Ifthis chemical has been inhaled, remove from exposure,begin rescue breathing (using universal precautions,including resuscitation mask) if breathing has stopped andCPR if heart action has stopped. Transfer promptly to amedical facility. When this chemical has been swallowed,get medical attention. If victim is conscious, administerwater or milk. Do not induce vomiting. The symptoms ofasthma may be delayed for several hours and are aggravatedby physical effort. Rest and medical observation are highlyrecommended. Medical observation is recommended for24°48 h after breathing overexposure, as pulmonary edemamay be delayed. As first aid for pulmonary edema, a doctoror authorized paramedic may consider administering a corticosteroid spray.
Color Code—White: Corrosive or Contact Hazard;Store separately in a corrosion-resistant location. Maleicanhydride must be stored to avoid contact with water andstrong oxidizers (such as chlorine and bromine), since violent reactions occur. Before entering confined space wherethis chemical may be present, check to make sure that anexplosive concentration does not exist. Sources of ignition,such as smoking and open flames, are prohibited where thischemical is handled, used, or stored. Metal containersinvolving the transfer of 5 gallons or more of this chemical should be grounded and bonded. Drums must be equippedwith self-closing valves, pressure vacuum bungs, and flamearresters. Use only nonsparking tools and equipment, especially when opening and closing containers of this chemical. Wherever this chemical is used, handled, manufactured,or stored, use explosion-proof electrical equipment andfittings.
UN2215 Maleic anhydride, Hazard class: 8; Labels: 8-Corrosive material.
Maleic Anhydride is commercialized and transported in the solid and molten forms. The molten Maleic Anhydride is transported at temperatures ranging from 60 to 80°C in well-insulated tank containers or road tankers provided with heating devices. In the solid form, it can be transported as pastilles, which are usually packed in polyethylene bags of 25 kg and transported either by rail tanker or by truck.
Crystallise it from *benzene, CHCl3, CH2Cl2 or CCl4. Sublime it under reduced pressure. [Skell et al. J Am Chem Soc 108 6300 1986, Beilstein 17 III/IV 5897, 17/11 V 55.]
Maleic anhydride was described as having anticarcinogenic
properties, and some of the maleic copolymers can have biologic
activity by themselves, especially antitumor activity.
Information related to this compound is contradictory. Chromosomal
aberrations in cultured hamster cells but no mutagenicity
in in vitro tests in bacteria have been reported.
No effects on cholinesterase activity have been described
after exposure to maleic anhydride.
Reacts slowly with water (hydrolyzes) to
form maleic acid, a medium-strong acid. Dust may form
explosive mixture with air. Reacts with strong oxidizers,
oil, water, alkali metals; strong acids; strong bases. Violent
reaction with alkali metals and amines above 66�C.
Dangerous reaction with oxidizers, amines, alkali metals,
and hydroxides. Compounds of the carboxyl group react
with all bases, both inorganic and organic (i.e., amines)
releasing substantial heat, water and a salt that may be
harmful. Incompatible with arsenic compounds (releases
hydrogen cyanide gas), diazo compounds, dithiocarbamates, isocyanates, mercaptans, nitrides, and sulfides
(releasing heat, toxic, and possibly flammable gases),
thiosulfates and dithionites (releasing hydrogen sulfate
and oxides of sulfur)
Consult with environmental
regulatory agencies for guidance on acceptable disposal
practices. Generators of waste containing this contaminant
(≥100 kg/mo) must conform to EPA regulations governing
storage, transportation, treatment, and waste disposal.
Controlled incineration: care must be taken that complete
oxidation to nontoxic products occurs.