Boron trifluoride is the inorganic compound with the formula BF3. It is a highly toxic, colorless and nonflammable gas with a penetrating and pungent odor. It dissolves quickly in water and any organic compounds containing nitrogen or oxygen. It can be slowly hydrolyzed by cold water to give off hydrofluoric acid, and can also hydrolyzes to form white dense fumes in moist air. Its vapors are heavier than air. Inhaling the gas will irritate the respiratory system and burns can result if the gas touches the skin in high concentrations.
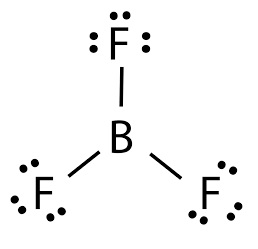
boron trifluoride lewis structure
Boron trifluoride is most importantly used as a reagent, typically as a Lewis acid, to catalyze such diverse operations as isomerization, alkylation, polymerization, esterfication, condensation, cyclization, hydration dehydration, sulfonation, desulfurization nitration, halogenation oxidation and acylation. Besides, it can also be used as a versatile building block for other boron compounds.
Boron trifluoride, BF3, is a colorless gas with a vapor density of 2.34, which is heavier than air. It is water-soluble and does not support combustion. It is also water-reactive, toxic by inhalation, and corrosive to skin and tissue. The TLV is 1 ppm, and the IDLH is 100 ppm in air. The boiling point is −148°F (64°C). The four-digit UN identification number is 1008. The NFPA 704 designation is health 4, flammability 0, and reactivity 1. The primary uses are as a catalyst in organic synthesis, in instruments for measuring neutron intensity, in soldering fluxes, and in gas brazing.
1. https://en.wikipedia.org/wiki/Boron_trifluoride
2. https://cameochemicals.noaa.gov/chemical/255
3. http://www.c-f-c.com/specgas_products/boron-trifluoride.htm
4.https://www.praxairdirect.com/Specialty-Gas-Information-Center/Pure-Gas-Specifications/Boron-Trifluoride.html
Boron trifluoride is a nonflammable, colorlessgas with a pungent, suffocating odor. Forms dense whitefumes in moist air. Shipped as a nonliquefied compressedgas. Molecular weight=67.8; Relative vapor density (air-5 1)=2.38; Boiling point=2100℃; Freezing/Meltingpoint=-127℃. Hazard Identification (based on NFPA-704 M Rating System): Health 3, Flammability 0,Reactivity 1 . Reacts with water; solubility in coldwater=106%.
Boron trifluoride is a nonflammable, colorless gas with an acrid suffocating odor. It forms thick acidic fumes in moist air. Dry boron trifluoride is used with mild steel, copper, copper-zinc and copper-silicon alloys, and nickel. Moist gas is corrosive to most metallic materials and some plastics. Therefore, Kel-F and Teflon are the preferred gasketing materials. Mercury containing manometers should not be used because boron trifluoride is soluble in mercury. It decomposes in hot water yielding hydrogen fluoride, Shipped as a nonliquefied compressed gas.
Colorless gas; pungent suffocating odor; density 2.975 g/L; fumes in moist air; liquefies at -101°C; solidifies at -126.8°; vapor pressure at -128°C is 57.8 torr; critical temperature -12.2°C; critical pressure 49.15 atm; critical volume 115 cm3/mol; soluble in water with partial hydrolysis; solubility in water at 0°C 332 g/100g; also soluble in benzene, toluene, hexane, chloroform and methylene chloride; soluble in anhydrous concentrated sulfuric acid.
Boron trifluoride is used as a catalyst for polymerizations, alkylations, and condensation reactions; To protect molten magnesium and its alloys from oxidation; as a gas flux for internal soldering or brazing; in ionization chambers for the detection of weak neutrons; and as a source of B10 isotope. By far the largest application of boron trifluoride is in catalysis with and without promoting agents.
Boron trifluoride is the
most widely used boron halide. Most applications take advantage of its strong Lewis acidity.
The most important uses are in organic syntheses.
Boron trifluoride is commonly used as a catalyst
for Friedel-Crafts alkylation reactions. It also is
used to catalyze the cleavage of ethers to alcohols, to catalyze esterification reactions, and in
the nitration and sulfonation of aromatic compounds. Many olefin polymerization reactions
use BF3 as an initiator, in conjunction with a
proton donor, such as water. Also BF3 is used to
catalyze the isomerization of alkenes and alkanes
and in petroleum cracking and desulfurization.
Amine complexes of BF3 are used as epoxy
curing agents.
ChEBI: Boron trifluoride is a boron fluoride.
Boron trifluoride is prepared by treating borax with hydrofluoric acid; or boric acid with ammonium bifluoride. The complex intermediate product is then treated with cold fuming sulfuric acid.
Boron trifluoride is a colorless gas with a pungent odor. Boron trifluoride is toxic by inhalation. Boron trifluoride is soluble in water and slowly hydrolyzed by cold water to give off hydrofluoric acid, a corrosive material. Its vapors are heavier than air. Prolonged exposure of the containers to fire or heat may result in their violent rupturing and rocketing.
Fumes in air. Soluble in water and slowly hydrolyzed by cold water to give hydrofluoric acid. Reacts more rapidly with hot water.
Boron trifluoride is a colorless, strongly irritating, toxic gas. Upon contact with water, steam or when heated to decomposition, Boron trifluoride will produce toxic fluoride fumes. Incompatible with alkyl nitrates, calcium oxide. Reaction with alkali metals or alkaline earth metals (except magnesium) will cause incandescence [Bretherick, 5th ed., 1995, p. 65].
Toxic by inhalation, corrosive to skin and
tissue. Lower respiratory tract irritant, and pneu-
monitis.
Boron trifluoride (and organic complexes such as BF3-etherate) are extremel corrosive substances that are destructive to all tissues of the body. Upon contact with moisture in the skin and other tissues, these compounds react to form hydrofluoric acid and fluoroboric acid, which cause severe burns. Boron trifluoride gas is extremely irritating to the skin, eyes, and mucous membranes. Inhalation of boron trifluoride can cause severe irritation and burning of the respiratory tract, difficult breathing, and possibly respiratory failure and death. Exposure of the eyes to BF can cause severe burns and blindness. This compound is not considered to have adequate warning properties. Boron trifluoride has not been found to be carcinogenic or to show reproductive or developmental toxicity in humans. Chronic exposure to boron trifluoride gas can cause respiratory irritation and damage.
When heated to decomposition or upon contact with water or steam, Boron trifluoride will produce toxic and corrosive fumes of fluorine containing compounds. Decomposes upon heating or on contact with moist air, forming toxic and corrosive fumes of boric acid and hydrofluoric acid. Reacts with alkalis and fumes in moist air, producing particulates which reduce visibility. Reacts with alkali metals, alkaline earth metals (except magnesium), alkyl nitrates, and calcium oxide. Boron trifluoride hydrolyzes in moist air to form boric acid, hydrofluoric acid, and fluoboric acid.
Flammability and Explosibility
Boron trifluoride gas is noncombustible. Water should not be used to extinguish any
fire in which boron trifluoride is present. Dry chemical powder should be used for
fires involving organic complexes of boron trifluoride.
Dry boron trifluoride does not react with the
common metals of construction, but If moisture
is present the acidic hydrates formed (BF3·H2O
and BF3·2H2O) can corrode many common metals
rapidly. Consequently, lines, pressure regulators,
and valves in boron trifluoride service
must be well protected from the entrance of
moist air between periods of use. Cast iron must
not be used because active fluorides attack its
structure. If steel piping is used for boron
trifluoride, forged-steel fittings must be used
instead of cast-iron fittings. Stainless steel, Monel,
nickel, and Hastelloy C are good materials
of construction.
Among materials suitable for gaskets are
Teflon, Kel F, and other appropriate fluorocarbon
or chlorofluorocarbon plastics. Most plastics
become embrittled in boron trifluoride
service. The use of polyvinyl chloride should be
avoided.
Boron trifluoride is a highly reactive
chemical used primarily as a catalyst in chemical synthesis.
It is stored and transported as a gas, but can be reacted
with a variety of materials to form both liquid and solid
compounds. The magnesium industry utilizes the fireretardant
and antioxidant properties of boron trifluoride
in casing and heat treating. Nuclear applications of boron
trifluoride include neutron detector instruments; boron-10
enrichment and the production of neutroabsorbing salts for
molten-salt breeder reactors.
Boron trifluoride irritates the nose, mucous
membranes, and other parts of the respiratory
system. Concentrations as low as I ppm in air
can be detected by the sense of smell and are
readily visible.
ACGIH recommends a Threshold Limit
Value-Ceiling (TLV-C) of 1 ppm (2.8 mg/m3)
for boron trifluoride. The TLV-C is the concentration
that should not be exceeded during
any part of the working exposure.
If contact with liquid, treat for frostbite. If thischemical gets into the eyes, remove any contact lenses atonce and irrigate immediately for at least 30 min, occasionally lifting upper and lower lids. Seek medical attentionimmediately. If this chemical contacts the skin, remove contaminated clothing and wash immediately with soap andwater. Seek medical attention immediately. If this chemicalhas been inhaled, remove from exposure, begin rescuebreathing (using universal precautions, including resuscitation mask) if breathing has stopped and CPR if heart actionhas stopped. Transfer promptly to a medical facility. Whenthis chemical has been swallowed, get medical attention.Give large quantities of water and induce vomiting. Do notmake an unconscious person vomit. Medical observation isrecommended for 24� 48 h after breathing overexposure, aspulmonary edema may be delayed. As first aid for pulmonary edema, a doctor or authorized paramedic may consideradministering a corticosteroid spray.
All work with boron trifluoride should be conducted in a
fume hood to prevent exposure by inhalation, and splash goggles and impermeable gloves
should be worn to prevent eye and skin contact. Cylinders of boron trifluoride should be
stored in locations appropriate for compressed gas storage and separated from alkali metals,
alkaline earth metals, and other incompatible substances. Solutions of boron trifluoride should
be stored in tightly sealed containers under an inert atmosphere in secondary containers.
UN1008 Boron trifluoride, Hazard class: 2.3;
Labels: 2.3—Poisonous gas, 8—Corrosive material,
Inhalation Hazard Zone B. Cylinders must be transported
in a secure upright position, in a well-ventilated truck.
Protect cylinder and labels from physical damage. The
owner of the compressed gas cylinder is the only entity
allowed by federal law (49CFR) to transport and refill
them. It is a violation of transportation regulations
to refill compressed gas cylinders without the express
written permission of the owner.
The usual impurities-bromine, BF5, HF and non-volatile fluorides-are readily separated by distillation. Brown and Johannesen [J Am Chem Soc 72 2934 1950] passed BF3 into benzonitrile at 0o until the latter was saturated. Evacuation to 10-5mm then removed all traces of SiF4 and other gaseous impurities. [A small amount of the BF3-benzonitrile addition compound sublimes and is collected in a U-tube cooled to -80o]. The pressure is raised to 20mm by admitting dry air, and the flask containing the BF3 addition compound is warmed with hot water. The BF3 that evolves is passed through a -80o trap (to condense any benzonitrile) into a tube cooled in liquid air. The addition compound with anisole can also be used. BF3 can be dried by passing it through H2SO4 saturated with boric oxide. It fumes in moist air. [It is commercially available as a 1.3M solution in MeOH or PrOH.] [Booth & Wilson Inorg Synth I 21 1939, Kwasnik in Handbook of Preparative Inorganic Chemistry (Ed. Brauer) Academic Press Vol I pp 219-222 1963.] TOXIC.
Boron trifluoride reacts with polymerized
unsaturated compounds. Decomposes on contact with
water, moist air, and other forms of moisture, forming toxic
and corrosive hydrogen fluoride, fluoroboric acid, and boric
acid. Reacts violently with alkali and alkaline earth metals
(except magnesium); metals, such as sodium, potassium,
and calcium oxide, and with alkyl nitrates. Attacks many
metals in presence of water.
Return refillable compressed
gas cylinders to supplier. The owner of the compressed gas
cylinder is the only entity allowed by federal law (49CFR)
to transport and refill them. Chemical reaction with water
to form boric acid, and fluoroboric acid. The fluoroboric
acid is reacted with limestone, forming boric acid and calcium
fluoride. The boric acid may be discharged into a sanitary
sewer system while the calcium fluoride may be
recovered or landfilled. Protect cylinder and labels from
physical damage.
Exposures to boron trifl uoride in occupational work areas cause irritating effects,
painful burns, lesions, and loss of vision. Workers with potential exposure to boron
trifl uoride should not wear contact lenses. Prompt medical attention is mandatory
in all cases of overexposure to boron trifl uoride and the rescue personnel should be
equipped with proper protectives. Occupational workers should handle/use boron trifl uoride only in well-ventilated areas. The valve protection caps must remain in place.
Workers should not drag, slide, or roll the cylinders, and use a suitable hand truck for
cylinder movement. Compressed gas cylinders shall not be refi lled without the express
written permission of the owner. Boron trifl uoride is listed as an extremely hazardous
substance (EHS).
The cylinder should not be heated by any means to increase the discharge rate of the
product from the cylinder. The cylinder of boron trifl uoride should be kept stored in a cool,
dry, well-ventilated area of non-combustible construction away from heavily traffi cked
areas and emergency exits
Boron trifluoride is available for commercial
and industrial use in technical grades having
much the same component proportions from one
producer to another.
Boron trifluoride is also available in
high-purity grades for use in the electronics
industry. Gas purity guidelines have been developed
and published by the Semiconductor
Equipment and Materials International and can
be found in the Book ofSEMI Standards, Gases
Volume.