Sodium pyrophosphate, also called Tetrasodium pyrophosphate or TSPP is used in the laboratory as a buffering agent. The compound has been shown to be useful in the preparation of an EDTA-sodium pyrophosphate extraction buffer for microcystin analysis of soil samples.
Tetrasodium pyrophosphate is an odorless, white powder or granules. It is used in water softener, buffering agent, thickening agent, dispersing agent, wool de-fatting agent, metal cleaner, soap and synthetic detergent builder, general sequestering agent, in electrodeposition of metals. It also acts as a tartar control agent in toothpaste and dental floss. In addition, it is used as a chelating agent in antimicrobial studies. It is also used as a food additive in common foods such as chicken nuggets, crab meat and canned tuna.
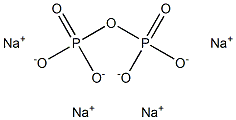
Tetrasodium pyrophosphate is a white crystalline powder or colourless crystals with the formula Na4P2O7. It contains the pyrophosphate ion and sodium cation. Toxicity is approximately twice that of table salt when ingested orally. There is also a hydrated form, Na4P2O7.10H2O.

Tetrasodium pyrophosphate is used as a buffering agent, an emulsifier, a dispersing agent, and a thickening agent, and is often used as a food additive. It is widely used as an emulsifying salt (ES) in process cheese. Common foods containing sodium pyrophosphate include chicken nuggets, marshmallows, pudding, crab meat, imitation crab, canned tuna, and soy-based meat alternatives and cat foods and cat treats where it is used as a palatability enhancer. It is the active ingredient in Bakewell, the substitute for baking powder's acid component marketed during shortages in World War II. It is also used in some common baking powders.
Pharmaceutic aid.
Tetrasodium pyrophosphate (Na4P2O7) is a non-toxic and biocompatible compound used as an electroactive media for exfoliation of the surface coating. It is also be used as an additive in the food industry.
Na4P2O7 can be used as an inorganic additive to improve the stability and electrochemical performance of redox flow batteries.
As a water softener; as a metal cleaner;
as a dispersing and emulsifying agent.
Tetrasodium Pyrophosphate is a coagulant, emulsifier, and
sequestrant that is mildly alkaline, with a ph of 10. it is moderately
soluble in water, with a solubility of 0.8 g/100 ml at 25°c. it is used
as a coagulant in noncooked instant puddings to provide thicken-
ing. it functions in cheese to reduce the meltability and fat separa-
tion. it is used as a dispersant in malted milk and chocolate drink
powders. it prevents crystal formation in tuna. it is also termed
sodium pyrophosphate, tetrasodium diphosphate, and tspp.
Tetrasodium pyrophosphate is formed when pure disodium hydrogen orthophosphate is heated to 500℃ for 5 hours. The product will contain better than 98 per cent Na4P2O7. Crystalline masses large enough for optical measurements are produced by heating in platinum to above the melting point of the tetrasodium pyrophosphate, 800°, and cooling slowly. Higher temperatures or longer heating times do not change the tetrasodium pyrophosphate, as this is the final product in the dehydration of disodium hydrogen orthophosphate.
ChEBI: Sodium diphosphate is an inorganic sodium salt comprised of a diphosphate(4-) anion and four sodium(1+) cations. More commonly known as tetrasodium pyrophosphate, it finds much use in the food industry as an emulsifier and in dental hygiene as a calcium-chelating salt. It has a role as a food emulsifier, a chelator and a food thickening agent. It contains a diphosphate(4-).
Odorless, white powder or granules. Mp: 995°C. Density: 2.53 g cm-3. Solubility in water: 3.16 g / 100 mL (cold water); 40.26 g / 100 mL boiling water. Used as a wool de-fatting agent, in bleaching operations, as a food additive. The related substance Tetrasodium pyrophosphate decahydrate (Na4P2O7 0H2O) occurs as colorless transparent crystals. Loses its water when heated to 93.8°C.
Tetrasodium pyrophosphate is basic. Reacts exothermically with acids. Incompatible with strong oxidizing agents. Decomposes in ethyl alcohol.
Tetrasodium pyrophosphate
(TSPP) is of low toxicity, but the dust may be
irritating to the eyes, upper respiratory tract,
and skin.
Mild to moderate skin and eye irritation
have occurred with acute exposure to the dust.
Poison by ingestion, intraperitoneal, intravenous, and subcutaneous routes. It is not a cholinesterase inhibitor. When heated to decomposition it emits toxic fumes of POx and Na2O.