Methanesulfonic acid (CH3SO3H, MSA) is a strong organic acid. The chemical oxidation of dimetyl sulfide in the atmosphere leads to the formation of MSA in large quantities. MSA undergoes biodegradation by forming CO2 and sulphate. It is considered a green acid as it is less toxic and corrosive in comparison to mineral acids.The aqueous MSA solution has been considered a model electrolyte for electrochemical processes.
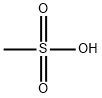
Methanesulfonic acid is an alkanesulfonic acid in which the alkyl group directly linked to the sulfo functionality is methyl. It has a role as an Escherichia coli metabolite. It is an alkanesulfonic acid and a one-carbon compound. It is a conjugate acid of a methanesulfonate.
Methanesulfonic acid, the simplest alkanesulfonic acid, is a colorless or slightly brown oily liquid, appearing as solid at low temperatures. It has a melting temperature of 20 °C, the boiling point of 167 °C (13.33 kPa), 122 °C (0.133 kPa), the relative density of 1.4812 (18 ℃) and refractive index 1.4317 (16 ℃). It is soluble in water, alcohol and ether, insoluble in alkanes, benzene and toluene. It will not subject to decomposition in boiling water and hot alkaline solution. It also has strong corrosion effect against the metal iron, copper and lead.
Methanesulfonic acid is a raw material for medicine and pesticide. It can also be used as dehydrating agent, curing accelerator for coating, treating agent for fiber, solvent, catalysis, and esterification as well as polymerization reaction.
It can be used as solvent, alkylation, catalyst of esterification and polymerization, also used in medicine and electroplating industry. It can also be applied to oxidation.
It can be obtained through the nitrate oxidation of thiocyanate methyl. Nitric acid and negative water are heated carefully to 80-88 °C with fractional addition of methyl thiocyanate and the temperature being automatically rose to about 105 ℃. After the reaction becomes mild, the reaction was heated to 120 ° C and reacted for 5 hours to obtain a crude product. The crude product was diluted with exchanged water and adjusted to pH 8-9 by addition of 25% barium hydroxide solution and filtered. The filtrate is condensed to until crystalline precipitation. The crystal is washed by methanol to remove the nitrate to obtain the barium methanesulfonate. It is then added to the exchanged water to boiling, add sulfuric acid for decomposition while it is hot, filter and the filtrate was concentrated under vacuum to no water to obtain the finished product.
Another method is that the methyl isothiourea sulfate is successively subject to chlorination, oxidation and hydrolysis to derive the finished product. Methyl isothiourea sulfate was added to the water; and the chlorine is sent into at 20-25 ° C to until phenomenon such as solution color is turned into yellow; oil layer emerges in the bottom of the bottle; the temperature drop and large number of residual chlorine is discharged from the exhaust pipe; this indicates the end point of the reaction. The reaction solution was extracted with chloroform. After drying, the extract was distilled at 60-62 ° C under normal pressure to remove the chloroform, and then further subject to distillation under reduced pressure. Collect the 60-65 °C (2.67 kPa) fraction was to obtain the methanesulfonyl chloride. Add the base drop wise under stirring to 80 ℃ hot water and maintain the heat hydrolysis for about 2h, to until the reaction liquid droplets completely disappear. The reaction solution was concentrated under reduced pressure to a syrupy form, diluted with water, and concentrated under reduced pressure to until no more water was distilled off to obtain methanesulfonic acid.
Methanesulfonic acid is a colourless or light yellow liquid having a melting point of 20° C, is a strong acid acting corroding but not oxidizing.
Methanesulfonic acid is used in the electroplating industry and for organic syntheses, in particular as a catalyst for alkylations, esterifications, and polymerizations. Beyond that, methanesulfonic acid is used as a starting material for the preparation of methanesulfonyl chloride.
Polymerization catalyst.
Methanesulfonic acid has been developed as an esterification catalyst in place of sulfuric acid for the synthesis of resins in paints and coatings. One of the major advantages of methanesulfonic acid over sulfuric acid is that it is not an oxidizing species.
Methanesulfonic acid is used as a catalyst in organic reactions namely esterification, alkylation and condensation reactions due to its non- volatile nature and solubility in organic solvents. It is also involved in the production of starch esters, wax oxidate esters, benzoic acid esters, phenolic esters, or alkyl esters. It reacts with sodium borohydride in presence of polar solvent tetrahydrofuran to prepare borane-tetrahydrofuran complex. It finds application in batteries, because of its purity and chloride absence. In pharmaceutical industry, it is used for the manufacturing of active pharmaceutical ingredients like telmisartan and eprosartan. It is useful in ion chromatography and is a source of carbon and energy for some gram-negative methylotropic bacteria.It is involved in the deprotection of peptides.
For complete protein and peptide hydrolysis with tryptophan recovery. After hydrolysis the samples are diluted prior to amino acid analysis.
ChEBI: An alkanesulfonic acid in which the alkyl group directly linked to the sulfo functionality is methyl.
Methanesulfonic acid is produced predominantly by oxidizing methylthiol or dimethyl disulfide using nitric acid, hydrogen peroxide, chlorine or by employing electrochemical processes.
Methanesulfonic acid (MSA) is a strong organic acid. The chemical oxidation of dimetyl sulfide in the atmosphere leads to the formation of MSA in large quantities. MSA undergoes biodegradation by forming CO2 and sulphate. It is considered a green acid as it is less toxic and corrosive in comparison to mineral acids.1 The aqueous MSA solution has been considered a model electrolyte for electrochemical processes.
Toxic by ingestion, skin irritant, corrosiveto tissue.
Poison by ingestion and
intraperitoneal routes. May be corrosive to
skin, eyes, and mucous membranes.
Explosive reaction with ethyl vinyl ether.
Incompatible with hydrogen fluoride. When
heated to decomposition it emits toxic
fumes of SOx. See also SULFONATES.
Dry the acid, either by azeotropic removal of water with *benzene or toluene, or by stirring 20g of P2O5 with 500mL of the acid at 100o for 0.5hours. Then distil it under vacuum and fractionally crystallise it by partial freezing. Sulfuric acid, if present, can be removed by prior addition of Ba(OH)2 to a dilute solution, filtering off the BaSO4 and concentrating under reduced pressure; and is sufficiently pure for most applications. [Beilstein 4 IV 10.]