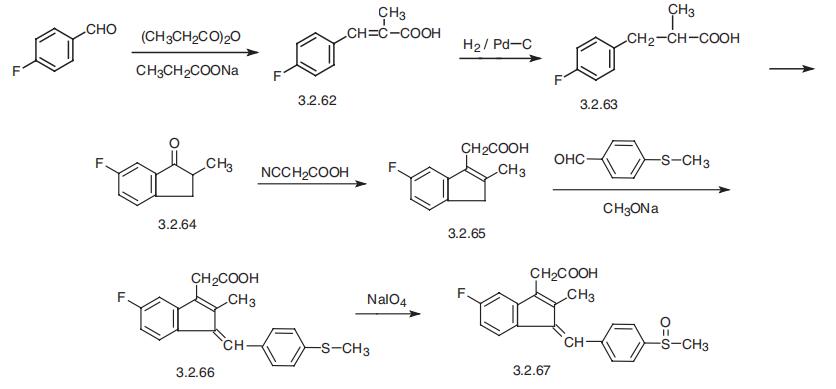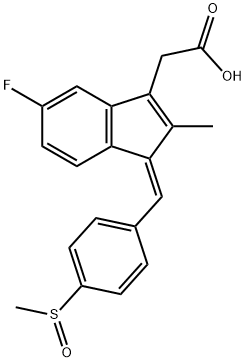
Sulindac
- Product NameSulindac
- CAS38194-50-2
- MFC20H17FO3S
- MW356.41
- EINECS253-819-2
- MOL File38194-50-2.mol
Chemical Properties
| Melting point | 182-185°C |
| Boiling point | 581.6±50.0 °C(Predicted) |
| Density | 1.2581 (estimate) |
| storage temp. | Sealed in dry,Store in freezer, under -20°C |
| solubility | Very slightly soluble in water, soluble in methylene chloride, sparingly soluble in ethanol (96 per cent). It dissolves in dilute solutions of alkali hydroxides. |
| pka | pKa (25°) 4.7 |
| color | Light yellow to Brown |
| Water Solubility | Soluble in water, methanol, ethanol. |
| λmax | 327nm(0.05mol/L methanolic HCl)(lit.) |
| Merck | 14,8982 |
| CAS DataBase Reference | 38194-50-2(CAS DataBase Reference) |
| NIST Chemistry Reference | Sulindac(38194-50-2) |
| EPA Substance Registry System | 1H-Indene-3-acetic acid, 5-fluoro-2-methyl-1-[[4-(methylsulfinyl)phenyl]methylene]-, (1Z)- (38194-50-2) |
Safety Information
| Hazard Codes | Xn |
| Risk Statements | 22-63-42/43 |
| Safety Statements | - |
| RIDADR | 3249 |
| WGK Germany | 3 |
| RTECS | NK8226000 |
| HazardClass | 6.1(b) |
| PackingGroup | III |
| HS Code | 29309090 |