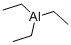The aluminum alkyls are highly flammable and reactive, colorless to yellow liquids at room temperature. The lighter trialkylaluminums ignite spontaneously in air. They are normally supplied and used in a 20% solution with a hydrocarbon solvent, such as hexane, heptane, benzene, toluene. Properties may depend on solvent. Reacts violently with water.
Colorless liquid. Miscible with saturated hydrocarbons.
Triethylaluminum is used as a co-catalyst in the industrial production of polyethylene and for the production of medium chain alcohols. Used as a catalyst in Ziegler-Natta polymerization process for vinyl, olefin, diene polymerizations and linear oligomerization and cyclization of unsaturated hydrocarbons. It is also used as a catalyst to produce ethylene gas, chain growth of ethylene, longer chain aluminum alkyls, and in plating aluminum.
Triethylaluminum, in combination withmany transition metal complexes, is used as Ziegler-Natta polymerization and hydrogenationcatalyst. Also, it is used as intermediatein organic syntheses.
Catalyst intermediate for polymerization of
olefins, especially ethylene; pyrophoric fuels; production
of α-olefins and long-chain alcohols; gas
plating of aluminum.
In a typical example, a slurry is prepared containing 7 wt % aluminum powder (0.1 wt% zirconium catalyst content) and 45 wt% recycled triethylaluminum in a paraffin hydrocarbon fraction, boiling range 177 – 260 ℃. The slurry is pumped into the reactor at a rate of 1.5 m3/h, at a reactor temperature of 132 ℃, while hydrogen is introduced at the bottom to maintain a partial pressure of 10 MPa. Excess hydrogen is removed in flash drum, wherein pressure is decreased to 3 MPa and temperature is lowered to prevent thermal reversal of the reaction. In the ethylation reactor sufficient ethylene is added to react completely with the Al – H bonds. Temperature is held at around 85 ℃ or somewhat higher. The addition rate increases with temperature, but carboalumination of ethylene also can occur to formn-butyl groups, especially at higher temperatures. Hydrogen and ethylene partial pressure is kept low in ethylation reactor to minimize hydrogenation of ethylene to ethane and to reduce excess carboalumination products. Excess ethylene and hydrogen are removed in flash drum. A portion of the triethylaluminum product solution is recycled to reactor.
A colorless liquid. Flammable gas is produced on contact with water.
Pyrophoric, ignites in moist air [Merck 11th ed. 1989]. Reacts violently with water.
Triethylaluminum reacts violently with water, alcohols, phenols, amines, carbon dioxide, sulfur oxides, nitrogen oxides, halogens, and halogenated hydrocarbons, causing fire and explosion hazards. [Handling Chemicals Safely 1980 p. 929]. A mixture of dimethylformamide and triethyl aluminum exploded when heated [Bretherick 1995].
Exposure to smoke from fire causes metal-fume fever (flu-like symptoms). Since liquid ignites spontaneously, contact with eyes or skin causes severe burns.
The health hazard from exposure to thiscompound is attributed to its violent reactionswith many substances, including airand water. Because of its violent reactionwith moisture, skin contact can cause a dangerousburn. Contact with eyes can damagevision.
Triethylaluminum is extremely pyrophoric,
igniting spontaneously in air. It reacts
violently with water, alcohol, halogenated
hydrocarbons, and oxidizing substances.
Among the alcohols, the lower alcohols,
methanol, ethanol, n- propanol, and
isopropyl alcohol, react explosively with
triethylaluminum. Reactions with lower
aldehydes, ketones and amides can be vigorous
to violent. It may explode on contact
with halocarbons in excess molar ratios or
upon slight warming. When heated to 200°C
(392°F), it decomposes, liberating ethylene
and hydrogen.
Flammability and Explosibility
Highly flammable
Extremely destructive
to living tissue. A very dangerous fire hazard
when exposed to heat or flame. Ignites
spontaneously in air. Explodes violently in
water. To fight fire, use CO2, dry sand, dry
chemical. Do not use water, foam, or
halogenated fire-fighting agents. Explosive
reaction with alcohols (e.g., methanol,
ethanol, propanol), carbon tetrachloride,
N,N-dmethylformamide + heat.
Incompatible with halogenated
hydrocarbons; triethyl borane. When heated
to decomposition it emits acrid smoke and
irritating fumes. See also ALUMINUM
COMPOUNDS and ORGANOMETALS.
Alkyl aluminum compounds are used as components of olefin polymerization catalysts. They are also used in the synthesis of higher primary alcohols and in pyrophoric fuels, as a catalyst in making ethylene gas; and in plating aluminum.
ntial fire or explosion hazard. Shipping: UN3399 Organometallic substance, liquid, water-reactive, flammable, Hazard Class: 4.3; Labels: 4.3
Dangerous Dangerous when wet material, 3-Flammable liquid, technical name Required. UN3051-Spontaneously combustible. Also, this material is dangerous when wet. (Note: this number does not appear in the 49/CFR HazMat tables).
Purify it by fractionation in an inert atmosphere under a vacuum in a 50cm column containing a heated nichrome spiral, taking the fraction b 112-114o/27mm. It is very sensitive to H2O and should be stored under N2. It should not contain chloride ions which can be shown by hydrolysis and testing with AgNO3. [Baker & Sisler J Am Chem Soc 75 4828 5193 1953, NMR: Brownstein et al. J Am Chem Soc 81 3826 1959, Beilstein 4 IV 4398.]
The lighter trialkylaluminums ignite spontaneously in air; can self-heat in the air at room temperature without any added energy and may ignite. These compounds are strong reducing agents. Incompatible with oxidizers (chlorates, nitrates, peroxides, permanganates, perchlorates, chlorine, bromine, fluorine, etc.); contact may cause fires or explosions. Keep away from alkaline materials, strong bases, strong acids, oxoacids, epoxides. Incompatible with water, oxygen (air), acids, alcohols, phenols, amines, carbon dioxide; sulfur oxides; halogenated compounds, and many other substances