HCFC 133a is odorless with poor warning properties.
HCFC133a is a nonisolated intermediate in the production of
halothane.
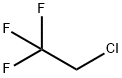
ChEBI: 2-Chloro-1,1,1-trifluoroethane is an organofluorine compound.
2-CHLORO-1,1,1-TRIFLUOROETHANE is a colorless, odorless gas. 2-CHLORO-1,1,1-TRIFLUOROETHANE is shipped as a liquid under its own vapor pressure. Contact with the liquid may cause frostbite to unprotected skin. 2-CHLORO-1,1,1-TRIFLUOROETHANE can asphyxiate by the displacement of air. Exposure of the container to fire or heat can cause 2-CHLORO-1,1,1-TRIFLUOROETHANE to rupture violently and rocket.
2-CHLORO-1,1,1-TRIFLUOROETHANE is chemically inert in many situations, but can react violently with strong reducing agents such as the very active metals and the active metals. Undergoes oxidation with strong oxidizing agents and under extremes of temperature.
Vapors may cause dizziness or asphyxiation without warning. Vapors from liquefied gas are initially heavier than air and spread along ground. Contact with gas or liquefied gas may cause burns, severe injury and/or frostbite. Fire may produce irritating, corrosive and/or toxic gases.
Some may burn but none ignite readily. Containers may explode when heated. Ruptured cylinders may rocket.
A poison by inhalation.
Experimental reproductive effects.
Questionable carcinogen with experimental
carcinogenic data. When heated to
decomposition it emits toxic fumes of F-.
HCFC 133a was evaluated in a
carcinogenesis screening study using Wistar-derived
rats. The rats were given daily intubations of 300mg HCFC
of body weight in corn oil for 52 weeks and held for a total of
125 weeks. In the males, there was a reduction in body
weight gain and testicular size, also there was an increase
in Leydig cell tumors and arrest of spermatogenesis.
In the females, there was an increased incidence of uterine
carcinomas.