colourless odourless gas (or colourless cryogenic liquid)
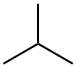
2-Methylpropane (isobutane), C4H10, a flammable gas,
occurs in small quantities in natural gas and crude oil. It
has been detected in urban atmospheres at concentrations of
44–74 ppb. It also evolves from natural sources and
has been measured in diesel exhaust at 1.4–11 ppm
and in cigarette smoke at 10 ppm. The partition
coefficient of propane between olive oil and air at 37℃ is
12 using the method described by Sato and Nakajima and
Perbellini et al.. The lower explosive limit is
18,000 ppm in air.
Isobutane occurs in petroleum, natural gas,and petroleum cracking products. It is usedas a fuel gas or a liquefied petroleum gas. Itis also used in organic synthesis.
In the production of propylene glycols
and oxides and polyurethane foams and resins;
as component of motor fuels and aerosol propellants;
as an industrial gas carrier and general
fuel source
Organic synthesis, refrigerant, motor fuels,
aerosol propellant, synthetic rubber, instrument calibration fluid.
ChEBI: An alkane that is propane substituted by a methyl group at position 2.
ISOBUTANE is a colorless gas with a faint petroleum-like odor. ISOBUTANE is shipped as a liquefied gas under its vapor pressure. Contact with the liquid can cause frostbite. ISOBUTANE is easily ignited. The vapors are heavier than air. Any leak can either be liquid or vapor. ISOBUTANE can asphyxiate by the displacement of air. Under prolonged exposure to fire or intense heat the containers may rupture violently and rocket.
ISOBUTANE is incompatible with the following: Strong oxidizers (e.g., nitrates & perchlorates), chlorine, fluorine, (nickel carbonyl + oxygen) .
Highly flammable, dangerous fire and
explosive risk; explosive limits in air 1.9–8.5%.
Central nervous system depression ranging from dizziness and incoordination to anesthesia and respiratory arrest, depending on concentration and extent of inhalation. Irregular heartbeat is rare but is a dangerous complication at anesthetic levels.
Isobutane, like other saturated aliphatic alkanes,is nontoxic. It is an asphyxiate. Exposureto high concentrations of 1% in air maycause narcosis and drowsiness. Other thanthis, there is no report of any adverse healtheffect from exposure to this gas.
EXTREMELY FLAMMABLE. Will be easily ignited by heat, sparks or flames. Will form explosive mixtures with air. Vapors from liquefied gas are initially heavier than air and spread along ground. CAUTION: Hydrogen (UN1049), Deuterium (UN1957), Hydrogen, refrigerated liquid (UN1966) and Methane (UN1971) are lighter than air and will rise. Hydrogen and Deuterium fires are difficult to detect since they burn with an invisible flame. Use an alternate method of detection (thermal camera, broom handle, etc.) Vapors may travel to source of ignition and flash back. Cylinders exposed to fire may vent and release flammable gas through pressure relief devices. Containers may explode when heated. Ruptured cylinders may rocket.
An asphyxiant. A
common air contaminant. A very dangerous
fire and explosion hazard when exposed to
heat, flame, or oxidizers. To fight fire, stop
flow of gas. When heated to decomposition
it emits acrid smoke and irritating fumes.
California Phase II reformulated gasoline contained 2-methylpropane at a concentration
of 1.04 g/kg. Gas-phase tailpipe emission rate from gasoline-powered automobiles equipped with
a catalytic converter was 130 μg/km (Schauer et al., 2002).
Photolytic. Based upon a photooxidation rate constant of 2.34 x 10-12 cm3/molecule?sec with OH
radicals in summer daylight, the atmospheric lifetime is 59 h (Altshuller, 1991). At atmospheric
pressure and 300 K, Darnall et al. (1978) reported a rate constant of 2.52 x 10-12 cm3/molecule?sec
for the same reaction. Rate constants of 1.28 x 10-9 and 6.03 x 10-12 L/molecule?sec were reported
for the reaction of 2-methylpropane with OH radicals in air at 300 and 296 K, respectively
(Greiner, 1967, 1970). Rate constants of 7.38 x 10-13 and 6.50 x 10-17 cm3/molecule?sec were
reported for the reaction of 2-methylpropane with OH and NO3, respectively (Sablji? and Güsten,
1990).
Chemical/Physical. Complete combustion in air produces carbon dioxide and water vapor. 2-
Methylpropane will not hydrolyze because it does not contain a hydrolyzable functional group.
(mole fraction):
In 1-butanol: 0.0897, 0.0491, and 0.0308 at 25, 30, and 70 °C, respectively; chlorobenzene: 0.157,
0.0837, and 0.0542 at 25, 30, and 70 °C, respectively; and octane: 0.301, 0.161, and 0.101 at 25,
30, and 70 °C, respectively (Hayduk et al., 1988).
In 1-butanol: 0.0889 and 0.0486 at 25 and 70 °C, respectively; in chlorobenzene: 0.162 and 0.0853
at 25 and 70 °C, respectively; and in carbon tetrachloride: 0.238 and 0.132 at 25 and 70 °C,
respectively (Blais and Hayduk, 1983).
Olefins and moisture can be removed by passage at 65o through a bed of silica-alumina catalyst which has previously been evacuated at about 400o. Alternatively, water and CO2 can be removed by passage through P2O5, then asbestos impregnated with NaOH. Treatment with anhydrous AlBr3 at 0o then removes traces of olefins. Inert gases can be separated by freezing the isobutane at -195o and evacuating out the system. [Beilstein 1 IV 282.]