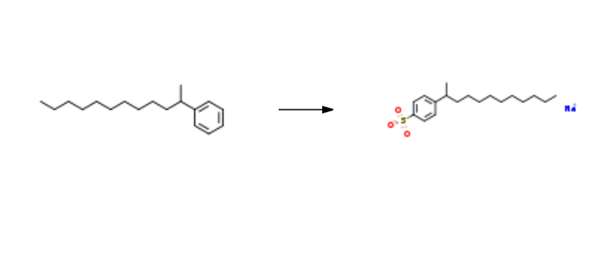
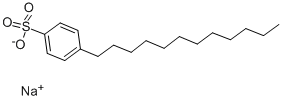
Sodium dodecylbenzenesulphonate is synthesised using 2-phenyldodecane as raw material by chemical reaction. The specific synthesis steps are as follows:
To a 5 L round bottom flask equipped with mechanical stirrer and an addition funnel was added octadecane and 2-phenyldodecane mixture ( 1.73 kg, 28%> 2-phenyldodecane). The reaction mixture was sparged with argon, warmed to 35°C and 1.25 weight % of oleum (632 g, 1.58 mol) was added dropwise, via addition funnel, to reaction mixture. The reaction mixture was stirred for 1.5 hours at room temperature. Upon completion, the reaction mixture was heated to 50°C and transferred to a separatory funnel and allowed to separate. The bottom layer was added slowly to a stirred solution of 15%> NaOH (aq) (2 L) at 10°C. Upon complete addition the resulting suspension was stirred for an additional 60 minutes. The solid was subsequently isolated by filtration and washed twice with ice-cold water. The solids were air dried for 16 hours and vacuum dried at 80°C to yield sodium, 4-(dodecan-2-yl) benzenesulfonate (555 g, 80.8% yield, 98.5% purity). lH NMR (400 MHz, (CD3)2S0/CDC13) δ 0.84 (t, J = 7.0 Hz, 3H), 0.95- 1.38 (m, 19H), 1.51 (pquart, J = 7.3 Hz, 2H), 2.65 (psext, J = 7.0 Hz, 1 H), 7.1 1 (d, J = 7.6 Hz, 2H), 7.56 (d, J = 8.4 Hz, 2H). 13C NMR (101 MHz, (CD3)2S0/CDC13) δ 13.8, 22.0, 22.1 , 27.0, 28.9, 28.9, 28.9, 28.9, 31.2, 37.6, 38.8, 125.4, 125.8, 145.3, 147.7.