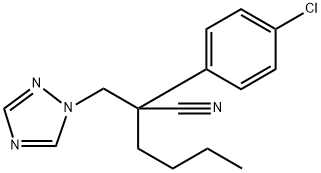
Myclobutanil is a broad-spectrum systemic foliar-applied
fungicide of the substituted triazole chemical class of
compounds, with protective, eradicative, and curative action. It
disrupts membrane function in fungi by inhibition of sterol
biosynthesis.
It is produced by the alkylation of 4-chlorophenylacetonitrile
with butyl chloride, followed by treatment with dibromomethane
and sodium hydroxide in dimethyl sulfoxide. The
resulting intermediate is then reacted with the potassium salt of
1,2,4-triazole in dimethyl formamide.
Commercially, myclobutanil is presented as different
formulations or preparations with concentrations up to 60%
active ingredient (a.i.). It is formulated as a wettable powder
(2–40% a.i.) or as an emulsifiable concentrate (1–25% a.i.),
granular (<1% a.i.), dust (5% a.i.), dry flowable (60% a.i.), and
ready to use (<1% a.i.) solutions to be used in agriculture or in
domestic activities by homeowners.
It is also used at lower concentrations in combination
with other agrochemicals such as pyretroids (permethrin),
fludioxonil, mefenoxam, azoxistrobin, mancozeb, sulfur, or
fluoxastrobin.
White to pale yellow crystalline solid. Soluble in alcohols, aromatics, esters, ketones and other organic solvents. Solubility is about 50-100g/L; insoluble in aliphatic hydrocarbons such as hexane; solubility in water is 142mg/L. The degradation half-life is 25 days in aqueous solution under sunlight, and 66 days in soil. It does not degrade under anaerobic conditions, and does not hydrolyze within 28 days under the conditions of pH 5, 7, and 9 (28°C).
Myclobutanil is an fungicide used on a wide range of food crops. Myclobutanil inhibits the biosynthesis of ergosterol, a critical componet of fungal cell membranes.
Myclobutanil is used for the control of Ascomycetes, Fungi
Imperfecti and Basidiomycetes on a wide variety of crops. These include
foliar treatment for scab and powdery mildew in apples and pears,
powdery mildew, shot hole, blossom blight, and rust in stone fruit,
powdery mildew in vines and cucurbits, powdery mildew and rusts on
ornamentals, rusts on perennial grasses grown for seed, and various diseases
of wheat. It is also used as a seed treatment for control of seed- and
soil-borne diseases in barley, maize, cotton, rice and wheat.
ChEBI: 2-(4-chlorophenyl)-2-(1H-1,2,4-triazol-1-ylmethyl)hexanenitrile is a nitrile that is hexanenitrile substituted at the 2-position by p-chlorophenyl and (1,2,4-triazol-1-yl)methyl groups. It is a nitrile, a member of triazoles and a member of monochlorobenzenes.
Moderately toxic by ingestion, inhalation,and skin contact.
Fungicide: Widely used to control powdery mildew, rust, sclerotina,
spot blight, rot, black rot and similar fungi on a variety
of food and non-food crops.
EAGLE®; NOVA®; NU-FLOW®;
RALLY®; LAREDO®; RH 3866®; SYSTHANE®
TECHNICAL
Moterately toxic by
ingestion, inhalation, and skin contact.
Experimental reproductive effects. When
heated to decomposition emits toxic fumes
of NOx, SO,, Cl-.
Myclobutanil is a nitrile compound
and a triazole fungicide. It is widely used to control powdery
mildew, rust, sclerotina, spot blight, rot, black rot and
similar fungi on a variety of food and nonfood crops.
Nitriles can be derived from salts of cyanide, but are usually
less toxic.
Relevant physicochemical properties are estimated Koc value
of 950, log Kow of 2.94, estimated Henry’s law constant of
4.3×10-9 atmm3 mol-1 at 25 ℃, vapor pressure of 1.6� 10�6
mmHg, and water solubility of 140 mg l-1.
In soil under aerobic conditions, myclobutanil exhibits high
to very high persistence, and medium to low mobility, forming
the minor soil metabolite myclobutanil butyric acid which
exhibits low to moderate persistence and very high mobility in
soil. Both adsorptions are affected significantly by differing soil
pH. Volatilization from moist soil surfaces is not expected to be
an important fate process. The biodegradation half-life of
myclobutanil in silt loam soil is about 66 days. No degradation
was observed under anaerobic conditions.
In aquatic environments, it is expected to adsorb to suspended
solids and sediment. Volatilization from water surfaces
is not expected. In dark natural sediment water systems,
myclobutanil partitioned from the water column to sediment,
where it exhibited very high persistence. Aqueous solutions of
myclobutanil decompose on exposure to light with half-lives of
222 days in sterile water, 0.8 days in sensitized sterile water,
and 25 days in pond water. Hydrolysis was not observed after
28 days at pH values of 5, 7, and 9 at 28 ℃.
An estimated bioconcentration factor of 37 suggests the
potential for bioconcentration in aquatic organisms is moderate,
provided the compound is not metabolized by the
organism.
In the ambient atmosphere, it is expected to exist in both
the vapor and particulate phases. Vapor-phase myclobutanil is degraded by reaction with photochemically produced hydroxyl
radicals; the half-life for this reaction in air is estimated to be
2.3 days. Particulate-phase myclobutanil may be removed from
the air by wet or dry deposition.
There is limited published information available on the metabolism of
myclobutanil. In plants and mammals, the butyl group is oxidised to an
alcohol and a ketone. In soils, substantial degradation of the molecule
occurs under aerobic conditions.
UN2763 Triazine pesticides, solid, toxic, Hazard
Class: 6.1; Labels: 6.1-Poisonous materials. UN3077
Environmentally hazardous substances, solid, n.o.s., Hazard
class: 9; Labels: 9-Miscellaneous hazardous material,
Technical Name Required. UN3082 Environmentally
hazardous substances, liquid, n.o.s., Hazard class: 9; Labels: 9-Miscellaneous hazardous material, Technical
Name Required.
Myclobutanil is stable under normal storage conditions (PM). Aqueous
solutions decompose on exposure to light. The DT50 is 222 days in sterile
water and 25 days in pond water. It was not hydrolysed in 28 days at pH
5,7 and 9 (28 °C).
Target organ in animal studies is the liver, as exposure to myclobutanil
resulted in liver hypertrophy or hepatotoxicity mediated
byCYP induction; also reproductivemodificationswere reported.
Triazole fungicides are designed to inhibit the fungal CYP51
enzyme but can also modulate the expression and function of
mammalian CYP genes and enzymes. Triazoles affected the
expression of numerous CYP genes in rat liver and testis,
including multiple CYP2C and CYP3A isoforms as well as other
xenobiotic metabolizing enzyme (XME) and transporter genes.
For some genes, myclobutanil and other triazoles had similar
effects on expression, suggesting possible common mechanisms
of action. Many of these CYP, XME, and transporter
genes are regulated by xenobiotic-sensing nuclear receptors,
and hierarchical clustering of CAR/PXR-regulated genes
demonstrated the similarities of toxicogenomic responses in
liver between all triazoles and in testis between myclobutanil
and triadimefon. Triazoles also affected expression of multiple
genes involved in steroid hormone metabolism in the two
tissues.
Conazole fungicides induce a thyroid response in rats.
Myclobutanil produced a dose-dependent decrease in T3 and
T4, related to an induction of uridine diphospho-glucuronosyl
transferase (UDPGT), the T4-metabolizing enzyme, although
thyroid hormone levels did not differ from control values after
90 days and TSH was not increased in any exposure group.
Decomposes on exposure to strong light.
Incompatible with oxidizers (chlorates, nitrates, peroxides,
permanganates, perchlorates, chlorine, bromine, fluorine,
etc.); contact may cause fires or explosions. Keep away from
alkaline materials, strong bases, strong acids, oxoacids, epoxides.
The triazoles are sensitive to heat, friction, and impact.
Sensitivity varies with the type substitution to the triazole
ring. Metal chelated and halogen substitution of the triazole
ring make for a particularly heat sensitive material. Azido
and nitro derivatives have been employed as high explosives.
No matter the derivative these materials should be treated as
explosives. Nitriles may polymerize in the presence of
metals and some metal compounds. They are incompatible
with acids; mixing nitriles with strong oxidizing acids can
lead to extremely violent reactions. Nitriles are generally
incompatible with other oxidizing agents such as peroxides
and epoxides. The combination of bases and nitriles can produce
hydrogen cyanide. Nitriles are hydrolyzed in both aqueous
acid and base to give carboxylic acids (or salts of
carboxylic acids). These reactions generate heat. Peroxides
convert nitriles to amides. Nitriles can react vigorously with
reducing agents. Acetonitrile and propionitrile are soluble in
water, but nitriles higher than propionitrile have low aqueous
solubility. They are also insoluble in aqueous acids.
Consult with environmental
regulatory agencies for guidance on acceptable disposal
practices. Contact a licensed disposal facility about surplus
and nonrecyclable solutions. Burn in a chemical incinerator
equipped with an afterburner and scrubber. Extra care must be exercised as the material in an organic solvent is highly
flammable. In accordance with 40CFR165, follow recommendations
for the disposal of pesticides and pesticide containers.
Containers must be disposed of properly by
following package label directions or by contacting your
local or federal environmental control agency, or by contacting
your regional EPA office. Incineration or permanganate
oxidation.