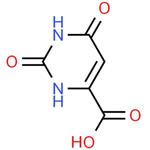
Orotic acid (also known as pyrimidinecarboxylic acid) is a heterocyclic acid; it is also known as. Historically it was believed to be part of the vitamin B complex and was called vitamin B13, but it is now known that it is not a vitamin. It is well known as a precursor in biosynthesis of pyrimidines; in mammals it is released from the mitochondrial dihydroorotate dehydrogenase (DHODH) for conversion to UMP by the cytoplasmic UMP synthase enzyme[1]. OA is also a normal part of the diet, being found in milk and dairy products[5], and it is converted to uridine for use in the pyrimidine salvage pathway predominantly in liver, kidney and erythrocytes. Before its essential role as an intermediate of pyrimidine biosynthesis was established in the 1950s[3], the compound OA was discovered in whey (Greek oros) by Biscaro and Belloni (1905)[4]. Since then it has received attention from several different aspects. For example, it is proposed that the dietary orotate is beneficial for animals and humans obviously arose from its considerable natural amounts in milk and dairy products[5]. In addition, it also draw great attention from its medical potentials such as improvement of learning behavior of adult rats, neuro-protective effect for gerbils and cats under transient cerebral ischemia, and optimization of functions in normal and ischemic rat hearts[6-9].
Orotate [orotic acid, OA] is the product of dihydroorotate dehydrogenase [DHODH], the fourth enzyme of pyrimidine [UMP] de novo synthesis [Fig. 1]. It is abundant in the serum and particularly urine of patients suffering from hereditary orotic academia or from enzyme defects in the urea cycle[10]. The sequence of six chemical reactions of UMP de novo synthesis is conserved from microorganisms to humans [Figure 1], although the organization of enzymes involved is different in eukaryotes[11-13]. In mammals, the six enzymes are coded by only three genes. The tri-functional CAD enzyme and the bifunctional UMP synthase each contain their appropriate catalytic activities on one polypeptide chain. The product of the third gene, dihydroorotate dehydrogenase [DHODH] holds a mitochondrial targeting sequence and a transmembrane domain at the N-terminus, which enable the enzyme to find its place in the inner mitochondrial membrane[14]. Through its electron acceptor, ubiquinone, DHODH is functionally united with the respiratory chain and can contribute to energy conservation in mitochondria. In turn, the formation of orotic acid [OA] by mitochondria, and hence the de novo synthesis of UMP, is strictly aerobic. In growth processes OA can be considered as a metabolic link between environmental oxygen tension and the proliferative capacity of cells[15].
Overview
Orotic Acid is used in the preparation of therapeutic agents for chronic obstructive pulmonary disease [COPD] treatment[16]. As it is an intermediate in de novo pyrimidine biosynthesis, it may be used to study the specificity and kinetics of orotate phosphoribosyltransferase [OPRT] which catalyzes the reversible phosphoribosyl transfer from 5′-phospho-α-d-ribose 1′-diphosphate [PRPP] to orotic acid [OA], forming pyrophosphate and orotidine 5′-monophosphate [OMP][17]. Orotic acid can also be used as a starting material for the potential commercial bio-production of uridine 5′-monophosphate [UMP] by microbes such as Corynebacterium ammoniagenes [ATCC 6872] or Saccharomyces cerevisiae. Moreover, it may be used to study the AMPK/SREBP-1 dependent cell-signaling pathway and transcription regulation mechanisms associated with the induction hepatic lipogenesis[17].
Medical aspects
OA has strong pharmacological potentials. The improvement of learning behavior of adult rats, neuro-protective effect for gerbils and cats under transient cerebral ischemia, and optimization of functions in normal and ischemic rat hearts were generally assigned to the precursor role of OA in cells for pyrimidine nucleotides and RNA[6-9]. The development of complexes and salts with metal ions [Mg2+, Zn2+, Ca2+, K+] and organic cations [e.g. choline, carnitine] exploited the favorable carrier function of OA for transportation and delivery of these ligands in living organisms to compensate deficiency syndromes[5]. Likewise, the construction of platinum, palladium and tin coordination compounds with OA for administration in cancer chemotherapy followed this principle[18]. The positive impact of OA preparations claimed for humans are highlighted by promises given in the public domain, such as beneficial cardiovascular effects, energy provision, improvement of body composition, and enhanced athletic performance.
Study has also shown that OA may lower the serum cholesterol and lipid[19-21]. Orotic acid also has strong uricosuric effect, originating from its role as a true transport substrate for the anion exchangerURAT1 [encoded by human SLC22A12 gene] located at the luminal site of the tubule cells[22].
Dietary aspects
The assumption that dietary orotate is beneficial for animals and humans obviously arose from its considerable natural amounts in milk and dairy products[5]. In the 1970s OA was proposed to increase the UDP-glucuronate pool for hepatic detoxification of bilirubin in the treatment of neonatal jaundice[5]. Another, underestimated, benefit of OA in the diet of newborns may be the establishment of the anaerobic OA-degrading intestinal microflora. Overall, it has strong potential to become a growth-enhancer for livestock.
Central Nervous systems
OA’s benefit for the brain, mentioned earlier, deserves closer attention. It can be assumed that a continuous supply of pyrimidine nucleotides is essential not only for the developing central nervous system[23], but equally for its plasticity, regeneration and neurotransmission: for example, some metabotropic P2Y receptor subtypes are sensitive to uridine nucleotides[24]. OA has putative protective and improvement effects of pyrimidine nucleotide pools, RNA synthesis, receptor saturation, repairing of ischeamia-induced membrane damage, which is explained by the function of OA as an intermediate in de novo UMP synthesis[25, 26].
Research field
OA had became a compound of considerable interest as a tool for studying pyrimidine metabolism in cells, tissues and animals: monitoring precursors, nucleotide pools and the rate of RNA synthesis, differentiating between de novo and salvage/recycling pathways, searching for antimetabolites and anticancer drugs[27, 29]. Concurrently, the growth-promoting features of OA were still of interest, and OA was used on animals and humans, with various biochemical rationales, assumptions and expectations[28, 30]. When OA was an additive to pharmaceutical preparations, its benefits were attributed to its being an intermediate of pyrimidine biosynthesis and therefore augmenting uridine nucleotide pools which are required for nucleic acid synthesis and for all pyrimidine nucleotide dependent biosynthetic processes[31, 32].
- https://www.springer.com/us/book/9780852002940
- Löffler, M.; Carrey E.; Zameitat, E. Orotic acid, more than just an intermediate of pyrimidine de novo synthesis, J. Genet. Genomics 2015, 42, 207–219.
- Reichard P.; Lagerkvist, U. The biogenesis of orotic acid in liver slices, Acta Chem. Scand. 1953, 7, 1207–1217.
- Biscaro, G.; Belloni E. About a new compound of the milk, Annuario della Soc. Chimica di Milano 1905, 11, 15.
- Löffler, M.; Carrey E.; Zameitat, E. Orotic acid,more than just an intermediate of pyrimidine de novo synthesis, J. Genet. Genomics 2015, 42, 207–219.
- Ru?thrich, H.; Wetzel, W.; Matthies, H. Postnatal orotate treatment: effects on learning and memory in adult rats, Psychopharmacology 1979, 63, 25–28.
- Krug, M.; Koch, M.; Schoof, E.; Wagner, M.; Matthies, H. Methylglucamine orotate, a memory-improving drug, prolongs hippocampal long-term potentiation, Eur. J. Pharmacol. 1989, 173, 223–226.
- Akiho, H.; Iwai, A.; Katoh-Sudoh,M.; Tsukamoto, S.; Koshiya, K.; Yamaguchi, T. Neuroprotective effect of Y-39558, orotic acid ethylester, in gerbil forebrain ischemia, Jpn. J. Pharmacol. 1998, 76, 441–444.
- Vilskersts, R.; Liepinsh, E.; Kuka, J.; Cirule, H.; Veveris, M.; Kalvinsh, I.; Dambrova, M. Myocardial infarct size-limiting and anti-arrhythmic effects of mildronate orotate in the rat heart, Cardiovasc. Drug Ther. 2009, 21, 281–288.
- Webster, D.R., Becroft, D.M., van Gennip, A.H., van Kuilenburg, A.B.P., 2001. Hereditary orotic aciduria and other disorders of pyrimidine metabolism. In: Scriver, C.R., Beaudet, A.L., Sly,W.S., Valle, D. [Eds.], The Metabolic and Molecular Bases of Inherited Disease, Vol. II. Medical Publishing Division, McGraw-Hill, New York, USA, pp. 2663e2702.
- Jones, M.E. Pyrimidine nucleotide biosynthesis in animals: genes, enzymes, and regulation of UMP biosynthesis, Ann. Rev. Biochem. 1980, 49, 253–279.
- Evans, D.R.; Guy, H.I. Mammalian pyrimidine biosynthesis: fresh insights into an ancient pathway, J. Biol. Chem. 2004, 279, 33035–33038.
- LöfflerM.; Zameitat, E.. Pyrimidine biosynthesis and degradation [catabolism] in The Encyclopedia of Biological Chemistry, eds. W.J. Lennarz; M.D. Lane, Academic Press, Waltham, 2013, Vol III, pp. 712–718.
- Rawls, J.; Knecht, W.; Diekert, K.; Lill, R.; Löffler, M. Requirements for the mitochondrial import and localization of dihydroorotate dehydrogenase, Eur. J. Biochem. 2000, 267, 2079– 2087.
- Löffler, M.; Carrey, E.A.; Zameitat, E. Essential role of mitochondria in pyrimidine metabolism, in Tumor Cell Metabolism: Pathways, Regulation and Biology, eds. S. Mazurek; S. Shoshan, Springer,Wien, Austria, 2015, pp. 287–312
- https://www.trc-canada.com/product-detail/?CatNum=O691500&CAS=50887-69-9%20&Chemical_Name=Orotic%20Acid%20Monohydrate&Mol_Formula=C?H?N?O?•H?O
- https://www.sigmaaldrich.com/catalog/product/sigma/o2750?lang=en®ion=US
- Nath, M.; Vats, M.; Roy, P. Tri- and diorganotin[IV] complexes of biologically important orotic acid: synthesis, spectroscopic studies, in vitro anti-cancer, DNA fragmentation, enzyme assays and in vivo anti-inflammatory activities, Eur. J. Med. Chem. 2013, 9, 310–321.
- Kelley,W.N.; Greene,M.L.; Fox, I.H.; Rosenbloom, F.M.; Levy R.I.; Seegmiller, J.E. Effects of orotic acid on purine and lipoprotein metabolism in man, Metabolism 1979, 19, 1025–1035.
- Robinson, J.L.; Dombrowski, D.B. Effects of orotic acid ingestion on urinary and blood parameters in humans, Nutr. Res. 1983, 3, 407–415.
- Robinson, J.K.;Dombrowski,D.B.; Tauss, L.R.; Jones, L.R.Assessment in humans of hypolipidemia induced by orotic acid, Am. J. Clin.Nutr. 1985, 41, 605–608.
- Miura,D.;Anzai,N.; Jutabha, P.; Chanluang, S.;He, X.; Toshiyuki, F.; Endou,H.Human urate transporter 1 [hURAT1] mediates the transport of orotate, J. Physiol. Sci. 2011, 61, 253–257.
- Connolly, G.P., Duley, J.A., 1999. Uridine and its nucleotides: biological actions, therapeutic potentials. Trends Pharmacol. Sci. 20, 218e225.
- Von Ku¨gelen, I., 2006. Pharmacological profiles of cloned mammalian P2Yreceptor subtypes. Pharmacol. Therapeut. 110, 414e432.
- Akiho, H., Iwai, A., Katoh-Sudoh, M., Tsukamoto, S., Koshiya, K., Yamaguchi, T., 1997. Post-ischaemic treatment with orotic acid prevented neuronal injury in gerbil brain ischaemia. Neuroreport 8, 607e610.
- Akiho, H., Iwai, A., Katoh-Sudoh, M., Tsukamoto, S., Koshiya, K., Yamaguchi, T., 1998a. Neuroprotective effect of Y-39558, orotic acid ethylester, in gerbil forebrain ischemia. Jpn. J. Pharmacol. 76, 441e444.
- Heidelberger, C., 1965. Fluorinated pyrimidines. Prog. Nucleic Acid Res. Mol. Biol. 4, 1e50.
- O’Sullivan, W.J., 1973. Orotic acid. Aust. N. Z. J. Med. 3, 417e423.
- Kaneti, J.J., Golovinsky, E.V., 1971. Quantitative relationships between the electronic structure and biological activity of some analogues of orotic acid. Chem. Biol. Interact. 3, 421e428.
- Falk, M., 1985. Orotsa¨ure. Die Pharmazie 40, 377e383
- Grisham, C.M., Garrett, R.H., Garrett, R., 2008. Biochemistry. Brooks Cole Publishing, Pacific Grove, USA.
- Voet, D., Voet, J.G., 2011. Biochemistry. John Wiley and Sons, New York, USA.
An intermediate in de novo pyrimidine biosynthesis.
hepatoprotectant, uricosuric agent
Orotic acid is an intermediate in de novo pyrimidine biosynthesis that may be used to study the specificity and kinetics of orotate phosphoribosyltransferase (OPRT) which catalyzes the reversible phosphoribosyl transfer from 5?-phospho-α-d-ribose 1?-diphosphate (PRPP) to orotic acid (OA), forming pyrophosphate and orotidine 5?-monophosphate (OMP). It is used as a starting material for the potential commercial bioproduction of uridine 5?-monophosphate (UMP) by microbes such as Corynebacterium ammoniagenes (ATCC 6872) or Saccharomyces cerevisiae. It may be used to study the AMPK/SREBP-1 dependent cell signaling pathway and transcription regulation mechanisms that induce hepatic lipogenesis.
ChEBI: A pyrimidinemonocarboxylic acid that is uracil bearing a carboxy substituent at position C-6.
91.7 g (0.5 mol) of trichloroacetyl chloride was cooled to -35°C in a glass vessel by means of a cooling brine. In the course of 3 h, 27.0 g (0.06 mol) of pure ketene was introduced through a tube. After completion of the reaction, the vessel was immediately put under dry nitrogen to prevent penetration of moisture. So γ,γ,γ-trichloroacetoacetylchloride was produced.
The reaction mixture containing the γ,γ,γ-trichloroacetoacetyl chloride was transfered under nitrogen to a dropping funnel and in the course of 15 min was added with vigorous agitation to a suspension of 69.0 g (1.15 mole) of urea in 90.0 g of anhydrous acetic acid. Water cooling was used so that the reaction temperature would not exceed 40°C. After completion of the addition, the reaction mixture was heated as rapidly as possible to 115°C, and held at
this temperature for 30 min.
Subsequently there was cooling and one more 99.0 g of glacial acetic acid and 180.0 g of water were added. The precipitated 6-trichloromethyluracil was filtered off and dried at 60°C in a vacuum drying cabinet. The yield was 91.0 g or 80%.
In a glass vessel equipped with an agitator, thermometer and pH electrode, 500 ml of water was placed and heated to 80°C. 50 g of 6trichloromethyluracil was then added. By means of the pH electrode, the addition of sodium hydroxide was automatically controlled so that the pH value throughout the whole hydrolysis was 6.5. Into, 165 ml of 5 N NaOH was consumed. Finally, the hydrolysis solution was cooled and the precipitated sodium orotate filtered off.
The crude sodium orotate was again suspended at 80°C in water and brought into solution (pH 10.5) by addition of 30 ml of 5 N NaOH. After treatment with active charcoal, the solution was acidified with 30.0 g of 50% sulfuric acid. The solution was then cooled. The orotic acid was filtered off and carefully washed with water. After drying, 20.5 g of orotic acid, having a purity of 99.3% (titration) was obtained. This corresponds to a 60% yield.
White crystals or crystalline powder.
Slightly soluble in water.
Carboxylic acids, such as Orotic acid, donate hydrogen ions if a base is present to accept them. They react in this way with all bases, both organic (for example, the amines) and inorganic. Their reactions with bases, called "neutralizations", are accompanied by the evolution of substantial amounts of heat. Neutralization between an acid and a base produces water plus a salt. Carboxylic acids with six or fewer carbon atoms are freely or moderately soluble in water; those with more than six carbons are slightly soluble in water. Soluble carboxylic acid dissociate to an extent in water to yield hydrogen ions. The pH of solutions of carboxylic acids is therefore less than 7.0. Many insoluble carboxylic acids react rapidly with aqueous solutions containing a chemical base and dissolve as the neutralization generates a soluble salt. Carboxylic acids in aqueous solution and liquid or molten carboxylic acids can react with active metals to form gaseous hydrogen and a metal salt. Such reactions occur in principle for solid carboxylic acids as well, but are slow if the solid acid remains dry. Even "insoluble" carboxylic acids may absorb enough water from the air and dissolve sufficiently in Orotic acid to corrode or dissolve iron, steel, and aluminum parts and containers. Carboxylic acids, like other acids, react with cyanide salts to generate gaseous hydrogen cyanide. The reaction is slower for dry, solid carboxylic acids. Insoluble carboxylic acids react with solutions of cyanides to cause the release of gaseous hydrogen cyanide. Flammable and/or toxic gases and heat are generated by the reaction of carboxylic acids with diazo compounds, dithiocarbamates, isocyanates, mercaptans, nitrides, and sulfides. Carboxylic acids, especially in aqueous solution, also react with sulfites, nitrites, thiosulfates (to give H2S and SO3), dithionites (SO2), to generate flammable and/or toxic gases and heat. Their reaction with carbonates and bicarbonates generates a harmless gas (carbon dioxide) but still heat. Like other organic compounds, carboxylic acids can be oxidized by strong oxidizing agents and reduced by strong reducing agents. These reactions generate heat. A wide variety of products is possible. Like other acids, carboxylic acids may initiate polymerization reactions; like other acids, they often catalyze (increase the rate of) chemical reactions.
Flash point data for Orotic acid are not available; however, Orotic acid is probably combustible.
Moderately toxic by ingestion, intraperitoneal, and intravenous routes. Mutation data reported. Whenheated to decomposition it emits toxic fumes of NOx.