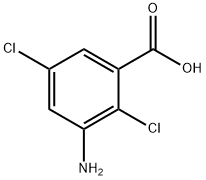
Chloramben is a colorless, odorless, crystallinesolid. Molecular weight=206.03; Freezing/Meltingpoint=200-201℃; Vapor pressure=6.8 3 1023 mmHg at100℃. Hazard Identification (based on NFPA 704 MRating System): Health 3, Flammability 0, Reactivity 0.Soluble in water; solubility=690 ppm at 25℃.
Off-white to beige powder
Chloramben is a colorless, odorless, crystalline solid.
Chloramben may be used as an analytical reference standard for the determination of the analyte in water samples using liquid chromatography with electrochemical detection (LC-EC).
Preemergence or preplant herbicide used in many vegetable and field crops to control
annual broad-leaved weeds and grasses. Also for postemergent control of common ragweed, redroot pigweed, smartweed and velvet-leaf.
Herbicide or plant growth regulator.
ChEBI: Chloramben is a chlorobenzoic acid.
Purplish white powder or light purple solid.
Chloramben reacts with sodium hypochlorite solutions .
ACUTE/CHRONIC HAZARDS: Chloramben emits toxic fumes when heated to decomposition.
Flash point data for Chloramben is not available, but Chloramben is probably combustible.
Herbicide: A herbicide for grasses, and broadleaf weeds. Mostly
used on soybeans, and also on corn, beans, asparagus, pumpkins,
peanuts, sunflowers, peppers, cotton, sweet potatoes,
squash, melons, hardwood trees, and some conifers. Not approved
for use in EU countries. Not registered for use in
the U.S. There are 51 global suppliers
ACP-M-728®; AMBEN®; AMBIBEN®;
AMIBEN®[C]; AMIBIN®; AMOBEN®; ORNAMENTAL
WEEDER®[C]; VEGABEN®; VEGIBEN®[C];
WEEDONE® GARDEN WEEDER
A General Use Pesticide (GUP) that is
no longer produced or sold in the United States. It is used
as an herbicide for grasses, broadleaf weeds, soybeans,
beans, and some vegetables. Workers involved in the manufacture, formulation, or application of this reemergence
herbicide.
If this chemical gets into the eyes, remove anycontact lenses at once and irrigate immediately for at least15 min, occasionally lifting upper and lower lids. Seekmedical attention immediately. If this chemical contactsthe skin, remove contaminated clothing and wash immediately with soap and water. Seek medical attention immediately. If this chemical has been inhaled, remove fromexposure, begin rescue breathing (using universal precautions, including resuscitation mask) if breathing hasstopped and CPR if heart action has stopped. Transferpromptly to a medical facility. When this chemical hasbeen swallowed, get medical attention. Give large quantities of water and induce vomiting. Do not make an unconscious person vomit.
Soil. In soils, chloramben was degraded by microorganisms but no products were
identified (Humburg et al., 1991). The main degradative pathway of chloramben in soil is
decarboxylation and subsequent mineralization to carbon dioxide. The calculated halflives in Ella loamy sand, Kewaunee clay and Poygan silty clay were 120–201, 182–286
and 176–314 days, respectively (Wildung et al., 1968). Persistence in soil is 6–8 weeks
(Hartley and Kidd, 1987).
Groundwater. According to the U.S. EPA (1986) and Ashton and Monaco (1991),
chloramben has a high potential to leach to groundwater, especially in sandy soils during
heavy rains.
Plant. Degrades in plants to N-glucoside, glucose ester, conjugates and insoluble
residues (Ashton and Monaco, 1991).
Photolytic. Plimmer and Hummer (1969) studied the irradiation of chloramben in water
(2–4 mg/L) under a 450-W mercury vapor lamp (λ >2,800 ?) for periods of 2 to 20 hours.
Chloride ion was released and a complex mixture of colored products was observed. It
was postulated that amino free radicals reacted with each other via polymerization and
oxidation processes. The experiment was repeated except the solution contained sodium
bisulfite as an inhibitor under a nitrogen atmosphere. Oxidation did not occur and loss of
the 2-chloro substituent gave 3-amino-5-chlorobenzoic acid (Plimmer and Hummer, 1969).
Chloramben (sodium salt) in aqueous solutions (100 mg/L) was rapidly photodegraded
in outdoor sunlight and under a 360-W mercury arc lamp (Crosby and Leitis, 1969).
In
sunlight, the solution became yellow-brown. Subsequent analysis by gas-liquid chromatography did not resolve any compounds other than chloramben. However, analysis by
TLC indicated at least 12 unidentified products. These products were reportedly formed
via replacement of chlorine by a hydroxy group, reductive dechlorination and abstraction
of hydrogen from the amine group (oxidation). No photodegradation products could be
identified in the solutions irradiated with the mercury arc lamp (Crosby and Leitis, 1969).
Chemical/Physical. Emits toxic fumes of nitrogen oxides and chlorine when heated
to decomposition (Sax and Lewis, 1987).
Forms water-soluble salts with alkalies.
Chloramben is generally stable to hydrolytic
degradation; however, it will decompose in sodium
hypochlorite solutions. It is also very sensitive to light
and under oxidative conditions aqueous solutions will
rapidly undergo photolysis. The predominant reaction in
oxidative photolysis is unclear but appears to involve the
formation of amino radicals. Photolysis also occurs under
reductive conditions by dechlorination at the 2 position,
i.e., when appropriate reducing agents are present, e.g.,
sodium bisulfite.
Color Code—Blue: Health Hazard/Poison: Storein a secure poison location. Prior to working with chloramben you should be trained on its proper handling and storage. Store in a cool, dry place, or a refrigerator, and avoidcontact with strong acids, acid fumes, and light. A regulated, marked area should be established where this chemical is handled, used, or stored in compliance with OSHAStandard 1910.1045.
UN2588 Pesticides, solid, toxic, Hazard Class:
6.1; Labels: 6.1-Poisonous materials, Technical Name
Required.
Although limited studies are
available, those that have been conducted, for example,
on cows and dogs, indicate that the majority of ingested
chloramben is quickly excreted through the urine and
feces and does not accumulate in tissues. An interesting
interaction between caffeine and chloramben suggests that
they can form an insoluble complex in the intestinal tract
and thus decrease the amount of chloramben absorbed
into the blood stream. The acute oral LD50 in rat is
>5000 mg/kg.
Rapidly decomposed by light. Strong
acids and acid fumes
Chloramben is stable to heat,
oxidation, and hydrolysis in acidic or basic media. The stability is comparable to that of benzoic acid. Wet oxidation
or incineration are recommended disposal methods.