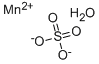Pale pink crystalline powder, slightly hygroscopic
Manganese(II) sulfate monohydrate is used as a colorant in dyes, fertilizers, animal feeds and red glazes on porcelain. Further, it is used in paints, ceramics, nutrient and dietary supplement. It is involved in the preparation of manganese dioxide. In addition, it serves as a precursor to manganese metal and other manganese compounds.
Manganese Sulfate is a source of manganese that functions as a nutrient and dietary supplement. It exists as a powder which is readily soluble in water.
Manganese sulfate monohydrate is used to produce manganese by an electrolytic process. The compound is used for dyeing textiles; for producing red glazes on porcelain; in varnish driers; in fertilizers; and in animal feeds to provide manganese as an essential trace element.
ChEBI: Manganese(II) sulfate monohydrate is a hydrate that is the monohydrate form of manganese(II) sulfate. It has a role as a nutraceutical. It is a hydrate, a manganese molecular entity and a metal sulfate. It contains a manganese(II) sulfate.
Manganese(II) sulfate is prepared by prolonged heating of any manganese salt with concentrated sulfuric acid. The compound is produced commercially from pyrolusite (MnO2) or rhodochrosite (MnCO3). Either mineral is dissolved in sulfuric acid and the solution evaporated:
MnO2 + H2SO4 → MnSO4 + H2O + ½ O2
Alternatively, manganese dioxide is heated strongly with dehydrated iron(II) sulfate:
2MnO2 + 2FeSO4 → 2MnSO4 + Fe2O3 + ½ O2
Manganese(II) sulfate, prepared by methods involving evaporation of manganese salt with sulfuric acid, is the tetrahydrate, MnSO4•4H2O. The tetrahydrate on gentle heating produces monohydrate, MnSO4•H2O.
Also, manganese(II) sulfate is a by-product in the manufacture of hydroquinone. The process involves reaction of aniline with manganese dioxide in sulfuric acid, followed by the removal of quinone by steam distillation under vacuum.
2C6H5NH2 (aniline) + 5H2SO4 + 4MnO2 → 2C6H4O2 + 4MnSO4 + 2C6H4O2 + 4MnSO4(quinone)
The unreacted acid is neutralized by lime, treated with water, and the solution filtered to separate any excess MnO2 and insoluble residues. Evaporation of the filtrate yields a crude product containing about 80% MnSO4 and 15% (NH4)2SO4.
Manganese(II) sulfate also may be produced by the action of sulfur dioxide with manganese dioxide:
MnO2 + SO2 → MnSO4
Odorless pale red slightly efflorescent crystals or light pink powder. pH (5% solution) 3.7.
Water soluble. Hygroscopic.
Manganese(II) sulfate hydrate is incompatible with aluminum and magnesium.
Flash point data for Manganese(II) sulfate hydrate are not available; however, Manganese(II) sulfate hydrate is probably combustible.
Manganese sulfate has various industrial applications such as dyeing, porcelain glazing, and the manufacture of fertilizers and boiling oils. MnSO4 is used as a source of manganese ion in biological research, such as in culturing of Bacillus licheniformis and the induction of chromosomal abnormalities in plants. MnSO4 has been utilized to investigate the enzyme dependent glycosylation of endogenous glycoproteins in human skeletal muscle.
Crystallise it from water (0.9mL/g) at 54-55o by evaporating about two-thirds of the water. It dehydrates above 400o.