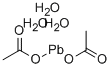
Lead acetate trihydrate, also known as plumbous acetate trihydrate, has the chemical formula Pb(C2H3O2)2·3H2O. It appears as a white, monoclinic crystalline solid. Upon heating it loses some of its water of crystallization, and after melting, decomposes at 200 °C. The trihydrate is highly soluble in water but insoluble in ethyl alcohol. It has an intensely sweet taste, hence it is sometimes called sugar of lead, but it is poisonous. The trihydrate is made by dissolving lead monoxide in hot dilute acetic acid solution; on cooling, large crystals separate, sometimes up to 60-cm long.
Lead(II) acetate trihydrate is used as a mordant in textile printing and dyeing, drier in paints, varnishes and as a water repellant. It is also used in cosmetics and to prepare other lead compounds. It serves as an ingredient in progressive types of hair coloring dyes. Further, it is used in the detection of poisonous gas hydrogen sulfide.
Lead acetate trihydrate, the usual commercial form, is used in the preparation of basic lead carbonate and lead chromate, as a mordant in cotton dyes, as a reagent for the manufacture of lead salts of higher fatty acids, as a water repellant, as a component in combined toning and fixing baths for daylight printing papers, and as a means of treating awnings and outdoor furniture to prevent removal of mildew- and rotproofing agents by rain or laundering. Other uses include preparation of rubber antioxidants; processing agent in the cosmetic, perfume, and toiletry industries; component of coloring agents for adhesives; and preparation of organic lead soaps as driers of paints and inks.
Lead(II) acetate trihydrate was a component of the solution which was used towards the fabrication of platinum black microelectrodes. Lead acetate trihydrate may be used in the manufacturing of other lead salts. It may be used in the textile industry for dyeing and printing. In paint industry, lead acetate acts as a drying agent and as a purification agent in chemical synthesis.
Lead acetate trihydrate appears as white powder or white chunky solid. Slight odor. pH (5% aqueous solution at 77°F) 5.5-6.5. It is water soluble and stable under normal conditions.
Slowly effloresces in air. Water soluble but solutions absorb carbon dioxide from the air and to give incompletely soluble products.
Lead acetate trihydrate may react with acids and oxidizing agents. Is incompatible with sulfates, citrates, tartrates, chlorides, carbonates, alkalis, tannin, resorcinol, salicylic acid, phenol, chloral hydrate, sulfites, vegetable infusions and tinctures. Incompatible with phosphates . Incompatible with KBrO3 and with EDTA.
Literature sources indicate that Lead acetate trihydrate is nonflammable.
Crystallise it twice from anhydrous acetic acid and dry it under vacuum for 24hours at 100o. The solubility in H2O is 63% (at ~20o) and 200% (at boiling point). [Beilstein 2 IV 118.]