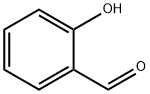
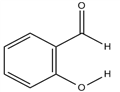
Salicylaldehyde (2-hydroxybenzaldehyde) is an organic compound with the formula C6H4CHO-2-OH. Along with 3-hydroxybenzaldehyde and 4-hydroxybenzaldehyde, it is one of the three isomers of hydroxybenzaldehyde. It is a colorless or pale yellow liquid with a bitter almond odor and a burning taste. It is soluble in alcohol, benzene, and ether, and very slightly soluble in water. Salicylaldehyde is found in shrubs of the genus Spiraea and is usually produced from phenol by the action of chloroform in the presence of an alkali base. It is used in the production of coumarin, saligenin, and salicylaldoxime (an important analytical reagent), and also in analytical chemistry—for example, to detect hydrazine. Besides, salicylaldehyde is a key precursor to various chelating agents and a flavouring ingredient.
Salicylaldehyde is a colourless to yellow oily liquid with a pungent, irritating, bitter, almond-like odor similar to benzaldehyde, acetophenone and nitrobenzene, but with phenolic notes. It has a nut-like, coumarin flavor at low levels. slightly soluble in water, soluble in ethanol and ether. It turns purple in case of ferric chloride.
Occurs frequently in nature; in the flowers of Spirea ulmaria and other Spireae, in the roots of Crepis foetida L., in the fruits of Pinus avium, in the rind of Rauqolfia caffra, in the leaves of Ceanothus velutinus and in the essential oil of Cinnamomum cassia and of tobacco leaves. Also reported found in grapes, tomato, baked potato, cinnamon bark, cassia leaf, peppermint oil, pennyroyal oil, parmesan cheese, butter, milk powder, roasted chicken, beer, rum, Japanese whiskey, sherry, coffee, tea, soybean, mushroom, buckwheat, Bourbon vanilla, Chinese quince, Muscat grape, vanilla and mastic gum oil.
Salicylaldehyde, is used as flavor and fragrance components. Salicylaldehyde is also a common highly-functionalized arene, that can be used as a precursor in the synthesis of other chemicals.
Salicylaldehyde is synthesized from phenol, chloroform, and alkali according to the Reimer–Tiemman method, which was developed in 1876. starting material for the manufacture of coumarin.
ChEBI: Salicylaldehyde is a hydroxybenzaldehyde carrying a hydroxy substituent at position 2. It has a role as a nematicide and a plant metabolite.
Taste characteristics at 20 ppm: spicy, medicinal and astringent
Liquid; colorless or pale yellow; bitter almond odor. Sinks and mixes slowly in water.
Salicylaldehyde is an aldehyde. Aldehydes are frequently involved in self-condensation or polymerization reactions. These reactions are exothermic; they are often catalyzed by acid. Aldehydes are readily oxidized to give carboxylic acids. Flammable and/or toxic gases are generated by the combination of aldehydes with azo, diazo compounds, dithiocarbamates, nitrides, and strong reducing agents. Aldehydes can react with air to give first peroxo acids, and ultimately carboxylic acids. These autoxidation reactions are activated by light, catalyzed by salts of transition metals, and are autocatalytic (catalyzed by the products of the reaction). The addition of stabilizers (antioxidants) to shipments of aldehydes retards autoxidation.
Salicylaldehyde is a skin irritant; 500 mg/daycaused moderate irritation to rabbit skin. Itcan have injurious effects on fertility. Studieson rats indicate that subcutaneous administrationof salicylaldehyde in a high doseof >400 mg/kg can produce developmentalabnormalities, fetal death, and postimplantationmortality.
The toxicity of this compound, however,is low. No toxic symptoms were noted.
LD50 value, oral (rats): 520 mg/kg
LD50 value, skin (rats): 600 mg/kg.
Combustible. Can react with oxidizing materials.
It is precipitated as the bisulfite addition compound by pouring the aldehyde slowly and with stirring into a 25% solution of NaHSO3 in 30% EtOH, then standing for 30minutes. The precipitate, after filtering at the pump, and washing with EtOH, is decomposed with aqueous 10% NaHCO3, and the aldehyde is extracted into diethyl ether, dried with Na2SO4 or MgSO4, and distilled, under reduced pressure. Alternatively, salicylaldehyde is precipitated as its Cu complex by adding it to warm, saturated aqueous Cu(OAc)2, shaking and standing in ice. The precipitate is filtered off, washed with EtOH, then Et2O, and decomposed with 10% H2SO4; the aldehyde is extracted into Et2O, dried and vacuum distilled. It was also purified by dry column chromatography on Kieselgel G [Nishiya et al. J Am Chem Soc 108 3880 1986]. The acetyl derivative has m 38-39o (from pet ether or EtOH) and b 142o/18mm, 253o/atm. [Beilstein 8 IV 176.] The oxime, [94-67-7] M 137.1, crystallises CHCl3/pet ether (b 40-60o) with m 57o [Beilstein 8 IV 203.]
Salicylaldehyde is burned in a chemicalincinerator equipped with an afterburner andscrubber.
- https://en.wikipedia.org/wiki/Salicylaldehyde
- https://pubchem.ncbi.nlm.nih.gov/compound/salicylaldehyde#section=Top
- http://www.wisegeek.com/what-is-salicylaldehyde.htm
- https://www.merriam-webster.com/medical/salicylaldehyde
- http://encyclopedia2.thefreedictionary.com/Salicylaldehyde