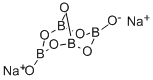Sodium borate, Na2B407.10H20, also known as sodium tetraborate and sodium pyrobomte, is a white crystalline powder that melts at 120°C (248 °F). Sodium borate in its natural impure form is also known as borax, Borax is a noncombustible (an inherent fire retardant), bluish-gray or green, odorless crystalline powder or granules. Sodium borate is used in glass and ceramic enamel mixes,detergents, fertilizers, pharmaceuticals, and photograph.
Refined borax (Na2B4O7) is an additive in laundry products such as soaps and water-softening
compounds. Also used for cosmetics, body powders, and the manufacture of paper and
leather. Borax is an environmentally safe natural herbicide and insecticide.
Sodium Tetraborate, is an important boron compound, which has a wide variety of applications. It is a component of many detergents, cosmetics, and enamel glazes. It is also used to make buffer solutions in biochemistry, as a fire retardant, as an anti-fungal compound for fiberglass, as a flux in metallurgy, neutron-capture shields for radioactive sources, a texturing agent in cooking, and as a precursor for other boron compounds.
Anhydrous borax is produced from borax through high
temperature fusion. On cooling, the clear, glass-like material
is ground into fine white granules. Because of its higher bulk
density, it is preferred where storage space is limited. It is
used principally in the manufacture of glass, ceramics, and
enamel.
Pale yellow solid or thick liquid with a faint odor of detergent. Mixes with water. Soap bubbles may be produced.
SODIUM PEROXOBORATE is incompatible with the following: Zirconium, strong acids, metallic salts . The true peroxoborate has been reported to detonate on light friction. The common "tetrahydrate" is not a peroxoborate, Sodium tetraborate is relatively stable under mild grinding with other substances.
No adverse effects from inhaling borax have been reported. Ingestion may cause acute or chronic effects; initial symptoms are nausea, vomiting, and diarrhea; these may be followed by weakness, depression, headaches, skin rashes, drying skin, cracked lips, and loss of hair; shock may follow ingestion of large doses and may interfere with breathing. Eye contact with powder or solutions may cause irritation; no chronic effects have been recognized, but continued contact should be avoided. Local skin irritation may result from contact with powder or strong solutions; the latter may cause chronic dermatitis on prolonged contact, and if skin is broken, enough boron may be absorbed to cause boron poisoning (symptoms are similar to those for ingestion).
Sodium tetraborate, also called borax, sodium borate, sodium pyroborate and sodium tetraborate decahydrate (Na2B4O7.10H2O) is a type of borate, and is used as a fertilizer to reduce boron deficiency. It is a white salt, finely ground for fertilizer application.
Sodium tetraborate(Na2B4O7) and sodium metaborate (Na2B2O4) are non-selective, taken up by roots, and have an unknown mechanism of action. Boron accumulates in reproductive structures after translocation from roots. Boron compounds are used for long-term, nonselective weed control in industrial and power line areas in combination with triazine and urea herbicides.
A nuisance dust.
Experimental reproductive effects. When
heated to decomposition it emits toxic
vapors of B.
Borax is used as a soldering flux,
preservative against wood fungus; and as an antiseptic.
Used in ant poisons, for fly control around refuse and
manure piles, as a larvicide. It is used in the manufacture
of enamels and glazes, fiberglass insulation; sodium perborate
bleach; in tanning, cleaning compounds; for fireproofing
fabrics and wood; and in artificial aging of
wood.
Most of the water of hydration is removed from the decahydrate (see below) by evacuation at 25o for three days, followed by heating to 100o and evacuation with a high-speed diffusion pump. The dried sample is then heated gradually to fusion (above 966o), allowed to cool gradually to 200o, then tranferred to a desiccator containing P2O5 [Grenier & Westrum J Am Chem Soc 78 6226 1956]. [Becher in Handbook of Preparative Inorganic Chemistry (Ed. Brauer) Academic Press Vol I pp 794-795 1963.]
Dissolves in water forming a basic solution.
Boron dust may form explosive mixture with air.
Contact with strong oxidizers may be violent. Boron is
incompatible with ammonia, bromine tetrafluoride, cesium
carbide, chlorine, fluorine, interhalogens, iodic acid, lead
dioxide, nitric acid, nitric oxide, nitrosyl fluoride, nitrous
oxide, potassium nitrite, rubidium carbide, silver fluoride.
Borax, dehydrated: The material
is diluted to the recommended provisional limit
(0.10 mg/L) in water. The pH is adjusted to between 6.5
and 9.1 and then the material can be discharged into sewers
or natural streams.