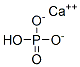
Dibasic Calcium Phosphate is anhydrous or contains two molecules of water of hydration. It occurs as a white, odorless, tasteless powder that is stable in air. It is practically insoluble in water, but is readily soluble in dilute hydrochloric and nitric acids. It is insoluble in alcohol.

Dibasic calcium phosphate is produced by the reaction of phosphoric acid, calcium chloride, and sodium hydroxide. Calcium carbonate can be used in place of the calcium chloride and sodium hydroxide.
Dibasic calcium phosphate anhydrous is generally regarded as relatively nontoxic and nonirritant material. It is widely used in oral pharmaceutical products and food products.
Functional Use in Foods: Leavening agent; dough conditioner; nutrient; dietary supplement; yeast food.
Dibasic calcium phosphate is found in nature as the mineral monetite. It is used as a food supplement and source of calcium, both in human food and animal feed. It is used in dough conditioner; in several dental products and in medicine. Other applications are in fertilizers, plastics and in the manufacture of glass.
Dibasic calcium hydrogen phosphate may be prepared by several methods.
It is precipitated by mixing solutions of calcium chloride and disodium hydrogen phospate:
CaCl2 + Na2HPO4 → CaHPO4 + 2NaCl
It also is prepared by treating phosphoric acid with lime water (suspension of calcium hydroxide in water). Also, it is obtained as a by-product in the preparation of hydroxypatite. The preparation involves the reaction of phosphoric acid with calcium phosphate.
H3PO4 + Ca3(PO4)2 → 3CaHPO4
Anhydrous dibasic calcium phosphate is a white, odorless, tasteless
powder or crystalline solid. It occurs as triclinic crystals.
White triclinic crystal; density 2.92 g/cm3 (anhydrous) and 2.31 g/cm3 (dihy drate); hardness 3.5 Mohs; decomposes on heating; inosluble in water and alcohol; KSP 2.7x10-7; soluble in dilute mineral acid.
It is used as a feed additive for livestock and poultry. It can promote feed digestion, put on animal?s weight, raise laying rate or producing milk rate, cure diseases such as rickets, osteomalacia and anemia.
Replenisher
(calcium); pharmaceutic aid (tablet base).
Calcium phosphates are usually prepared by reacting very pure
phosphoric acid with calcium hydroxide, Ca(OH)2 obtained from
limestone, in stoichiometric ratio in aqueous suspension followed
by drying at a temperature that will allow the correct hydration
state to be achieved. After drying, the coarse-grade material is
obtained by means of a classification unit; the fine particle-size
material is obtained by milling. Dibasic calcium phosphate,
anhydrous, may also be prepared by spray-drying.
ChEBI: Calcium hydrogenphosphate is a calcium phosphate.
CalStar (FMC); D.C.P. (Parke-Davis).
Flammability and Explosibility
Non flammable
Dicalcium phosphate (CaHPO4) is made from calcium carbonate and phosphoric acid. It contains 34% citrate- soluble phosphorus pentoxide (P2O5). It is not commonly used as a fertilizer, but is used as a supplement to animal feed.
Pharmaceutical Applications
Anhydrous dibasic calcium phosphate is used both as an excipient
and as a source of calcium in nutritional supplements. It is used
particularly in the nutritional/health food sectors. It is also used in
pharmaceutical products because of its compaction properties, and
the good flow properties of the coarse-grade material. The
predominant deformation mechanism of anhydrous dibasic calcium
phosphate coarse-grade is brittle fracture and this reduces the
strain-rate sensitivity of the material, thus allowing easier transition
from the laboratory to production scale. However, unlike the
dihydrate, anhydrous dibasic calcium phosphate when compacted
at higher pressures can exhibit lamination and capping. This
phenomenon can be observed when the material represents a
substantial proportion of the formulation, and is exacerbated by the
use of deep concave tooling. This phenomenon also appears to be
independent of rate of compaction.
Anhydrous dibasic calcium phosphate is abrasive and a lubricant
is required for tableting, for example 1% w/w magnesium stearate
or 1% w/w sodium stearyl fumarate.
Two particle-size grades of anhydrous dibasic calcium phosphate
are used in the pharmaceutical industry. Milled material is typically
used in wet-granulated or roller-compacted formulations. The
‘unmilled’ or coarse-grade material is typically used in directcompression
formulations.
Anhydrous dibasic calcium phosphate is nonhygroscopic and
stable at room temperature. It does not hydrate to form the
dihydrate.
Anhydrous dibasic calcium phosphate is used in toothpaste and
dentifrice formulations for its abrasive properties.
Skin and eye irritant. A
nuisance dust.
Dibasic calcium phosphate anhydrous is widely used in oral
pharmaceutical products, food products, and toothpastes, and is
generally regarded as a relatively nontoxic and nonirritant material.
Dibasic calcium phosphate anhydrous is a nonhygroscopic,
relatively stable material. Under conditions of high humidity it
does not hydrate to form the dihydrate.
The bulk material should be stored in a well-closed container in a
dry place.
Crystallise it from a near-saturated solution in 50% aqueous reagent grade phosphoric acid at 100o by filtering through fritted glass and cooling to room temperature. The crystals are filtered off, and this process is repeated three times using fresh acid. For the final crystallisation the solution is cooled slowly with constant stirring to give thin plate crystals that are filtered off on a fritted glass funnel, washed free of acid with anhydrous acetone and dry in a vacuum desiccator [Egan et al.J Am Chem Soc 78 1811 1956].
Dibasic calcium phosphate should not be used to formulate
tetracyline antibiotics.
The surface of milled anhydrous dibasic calcium phosphate is
alkaline and consequently it should not be used with drugs that
are sensitive to alkaline pH. However, reports suggest there are
differences in the surface alkalinity/acidity between the milled and
unmilled grades of anhydrous dibasic calcium phosphate; the
unmilled form has an acidic surface environment. This difference
has important implications for drug stability, particularly when
reformulating from, e.g. roller compaction to direct compression,
when the particle size of the anhydrous dibasic calcium phosphate
might be expected to change.
Dibasic calcium phosphate dihydrate has been reported to be
incompatible with a number of drugs and excipients, and many of
these incompatibilities are expected to occur with dibasic calcium
phosphate, anhydrous; see Calcium phosphate, dibasic dihydrate.
GRAS listed. Accepted as a food additive in Europe. Included in the
FDA Inactive Ingredients Database (oral capsules and tablets).
Included in nonparenteral medicines licensed in Europe. Included in
the Canadian List of Acceptable Non-medicinal Ingredients.