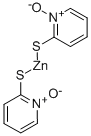
Zinc pyrithione is the pyrithione complexes of zinc bromide, in the early 1930s, was already synthesized and used as a topical antifungal or antibacterial agent. At room temperature it is white to yellow crystalline powder. Slight characteristic odor. Insoluble in water. Solubility: water 15mg/kg; pH = 8 Water 35mg/kg; Ethanol 100mg/kg; polyethylene glycol (PEG400) 2000mg/kg. A pH optimum range of 4.5 to 9.5; mass fraction of 10% suspension pH3.6. Zinc pyrithione react with cationic and non-ionic surfactants forming insoluble precipitate, unstable in the light and oxidizer, when at higher temperatures ,it is not stable to acids and bases . It is not compatible with EDTA, non-ionic surfactants make it partially deactivated. When with the presence of heavy metals, chelation or anti-sequestration will occur, and these chelates are insoluble in water.
EEC and GB7916-87 provide that maximum allowable concentration of mass fraction of zinc pyrithione on cosmetics is 0.5%, only for cleaning after using products.General concentration 250~1000mg/kg (active), zinc pyrithione can be used in gels, creams, lotions, talcum powder and anti-dandruff shampoo, deodorant and also for disinfecting articles.
Shampoo for dandruff,zinc pyrithione can inhibit Gram positive and negative bacteria and mold growth,Care hair Effectively , delay hair aging ,control white hair and hair loss generation. zinc pyrithion is also used as a cosmetic preservative, oil, paint biocide.
Zinc pyrithione has a strong killing power on fungi and bacteria so that it can effectively kill dandruff fungus,playing a role in dandruff .
Pharmaceutical Applications
Zinc pyrithione is a regulator of keratinization, selenium sulfide has antimicrobial properties and ketoconazole is an antifungal agent (i.e.can provide the reduction of the lipophilic yeast Malassezia furfur).
Zinc pyrithione shampoos (in concentrations between 1 to 2%) and shampoos with selenium sulide have been reported to be effective in the treatment of scalp psoriasis.However,their efficacy has not been substantiated by controlled studies.There is also some evidence that shampoos with antifungal agents (e.g.ketoconazole)can have a beneficial effect on scalp conditions. It should be noted, however, that the effectiveness of these shampoos is much smaller than these containing tars, corticosteroids or keratolytics.
Zinc pyrithione is very rapidly transformed by photolysis. Experiments conducted under sterile conditions with a light:dark cycle of 12:12 hours have shown that, under exposure to light, the concentration of [pyridine-2,6-14C]zinc pyrithione in pH 9 buffer was reduced to 33% of the radioactivity added in 15 min. Data from this study also demonstrated that less than 5% of the 14C added occurred as zinc pyrithione after 1 hour of exposure to light. Similar results have been achieved when photolysis of zinc pyrithione was investigated by use of artificial seawater. In this study, the parent compound constituted 45% of the radioactivity added after 15 min while, after 24 hours, 1.3% of the added dose occurred as zinc pyrithione. The estimated half-lives of the photolytic transformation of zinc pyrithione was 13 min in pH 9 buffer and 17.5 min in artificial seawater (Reynolds 1995a).
https://www2.mst.dk/udgiv/publications/2000/87-7944-084-3/html/kap04_eng.htm
The toxicity of the active substance zinc pyrithione has been investigated in standard laboratory tests with a number of aquatic organisms living in fresh water (the green alga Selenastrum capricornutum, the crustacean Daphnia magna, the fish rainbow trout (Oncorhynchus mykiss) and fathead minnow (Pimephales promelas)) and in seawater (the crustacean Mysidopsis bahia, the fish sheepshead minnow (Cyprinodon variegatus) and the oyster (Crassostrea virginica)) (Boeri et al. 1993; 1994a-e; Ward et al. 1994a).
The results show that while zinc pyrithione and omadine disulfide were very toxic to aquatic organisms (L(E)C50 in the order of 3-300 µg/L), omadine sulfonic acid and pyridine sulfonic acid were considerably less toxic (L(E)C50 in the order of >20 mg/L) (Olin 1977). In a long-term study with fish eggs and larvae, pyridine sulfonic acid gave no effects at a concentration of 0.01 mg/L (Boeri et al. 1999). Algae were the group of organisms most sensitive to the last two substances.
Zinc pyrithione is a coordination complex of zinc and pyrithione that has antimicrobial and anticancer activities. It is active against the bacteria E. coli, S. aureus, K. pneumoniae, A. baumannii, P. aeruginosa, E. faecium, E. faecalis, and E. cloacae (MICs = 1-4 μg/ml) and the fungus P. ovale when used at concentrations ranging from 0.01 to 10 μg/ml. Zinc pyrithione reduces tumor growth in an SCC-4 mouse xenograft model when administered at a dose of 1 mg/kg per week for six weeks. Formulations containing zinc pyrithione have been used in the treatment of dandruff.
Reactions may lead to photosensitive eczema and actinic reticuloid syndrome. Zinc pyrithione is used as antifungal, antibacterial and antiseborrheic agent used in many shampoos and hair creams.
zinc pyrithione is a preservative against bacteria, fungi, and yeast. It is unstable in light and in the presence of oxidizing agents. Zinc pyrithione is useful in gels, creams, heavy lotions, and talcum powder.
Zinc pyrithione is directly cytotoxic and has antimicrobial effects. Zinc pyrithione is found in many shampoos (DHS-Zinc, Head and Shoulders) and should be applied for 5 minutes daily for 2 weeks.
Zinc pyrithione is the active ingredient in several shampoos used to control dandruff and seborrheic dermatitis and is also effective in the therapy of tinea versicolor. It remains unclear whether the beneficial effects are caused by an antiproliferative or antimicrobial effect or both. It is substantive to the hair, allowing continued therapeutic effect after washing.
Zinc pyrithione is the active ingredient in several shampoos used to control
dandruff and seborrheic dermatitis and is also effective in the therapy of tinea
versicolor. It remains unclear whether the beneficial effects are caused by an
antiproliferative or antimicrobial effect or both. It is substantive to the hair,
allowing continued therapeutic effect after washing.
Head & Shoulders Conditioner (Procter & Gamble).
Organometallics, such as Zinc pyrithione, are reactive with many other groups. Incompatible with acids and bases. Organometallics are good reducing agents and therefore incompatible with oxidizing agents. Often reactive with water to generate toxic or flammable gases.
Flash point data for Zinc pyrithione are not available, but Zinc pyrithione is probably combustible.
Zinc pyrithione is widely used in antidandruff
shampoos and is a classic allergen. Concomitant reactions
are expected to both zinc and sodium pyrithione.
Poison by ingestion,
skin contact, intraperitoneal, and
intravenous routes. Moderately toxic by
subcutaneous route. An experimental
teratogen. Experimental reproductive
effects. An eye irritant. When heated to
decomposition it emits very toxic fumes of
NOx, SOx, and ZnO. Used as an anti-
dandruff agent in shampoos. See also ZINC
COMPOUNDS and SULFIDES.
1000 g of sodium pyrithione, 20% aqueous solution (1.34 mmol of sodium pyrithione) was added to a 2L reactor equipped with a mechanical agitator and warmed to 70 °C with stirring. To the solution was added 190 g of 50 % zinc chloride (0.7 mmol of zinc chloride) over 10 minutes. After the completion of the addition, the solution was stirred at 70 °C for one hour, dehydrated and then washed with 500 g of distilled water. The solid thus obtained was dried with a heated-air drier at 70 °C to have a moisture content of less than 0.5 % and ground with a hammer mill to obtain a find powder of zinc pyrithione. The zinc pyrithione thus obtained was 212 g with a yield of 99.5 %.