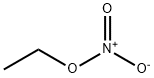
Ethyl nitrate has formula C2H5NO3. It is used in organic synthesis and as an intermediate in the preparation of some drugs, dyes, and perfumes.
Ethyl nitrate is found in the atmosphere, where it can react with other gases to form smog. Originally thought to be a pollutant, formed mainly by the combustion of fossil fuels, recent analysis of ocean water samples reveal that in places where cool water rises from the deep, the water is saturated with alkyl nitrates, likely formed by natural processes.
Colorless liquid; pleasant odor; sweet
taste.Soluble in alcohol and ether;
insoluble in water.
Ethyl nitrate is capable of explosive decomposition when exposed to heat.
Organic synthesis, drugs, perfumes, dyes,
rocket propellant.
The alkyl nitrates are produced from the corresponding alcohols by esterification
with nitric acid in the presence of urea or urea nitrate.
Ethyl nitrate has been prepared by bubbling gaseous nitryl fluoride through ethanol at ?10 °C . The reaction was subsequently studied in detail.
A clear colorless liquid with a pleasant odor. Flash point 50°F. Prolonged exposure to fire or heat may cause vigorous decomposition and rupturing of the container. Denser than water and insoluble in water. Vapors are heavier than air. Produces toxic oxides of nitrogen during combustion.
Highly flammable. Insoluble in water.
Organonitrates, such as Ethyl nitrate, range from slight to strong oxidizing agents. If mixed with reducing agents, including hydrides, sulfides and nitrides, they may begin a vigorous reaction that culminates in a detonation. Nitroalkanes are milder oxidizing agents, but still react violently with reducing agents at higher temperature and pressures. Nitroalkanes react with inorganic bases to form explosive salts. The presence of metal oxides increases the thermal sensitivity of nitroalkanes. Nitroalkanes with more than one nitro group are generally explosive.
Flammable, dangerous, fire and explosion
risk.
Inhalation or contact with material may irritate or burn skin and eyes. Fire may produce irritating, corrosive and/or toxic gases. Vapors may cause dizziness or suffocation. Runoff from fire control or dilution water may cause pollution.
In humans ethyl nitrate can cause headache, narcosis and vomiting, but no cases of
industrial intoxication have been reported .
Ethyl nitrate is used in the synthesis of certain drugs, perfumes and dyes. It has
also found some use as a rocket fuel.
A poison by
intraperitoneal route. Mutation data
reported. A very dangerous fire hazard when
exposed to heat or flame; can react
vigorously with oxibzing materials. A
moderate explosion hazard when exposed to
heat (explodes @ 185'F). To fight fire, use
foam, CO2, dry chemical, water to blanket
fire. Incompatible with Lewis acids. When
heated to decomposition it emits toxic
fumes of NOx. See also NITRATES and ESTERS.
There is no information concerning the in vivo metabolism of ethyl nitrate, but it
seems likely that one route for biotransformation is hydrolysis to yield an alcohol
and nitrate. Such a pathway is common to other alkyl nitrates such as nitroglycerin
and amyl nitrate.