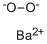Barium peroxide, is a grayish-white powder that is slightly soluble in water. Barium peroxide is a dangerous fire and explosion risk in contact with organic materials and decomposes around 1450°F (787°C). It is also toxic by ingestion, is a skin irritant, and should be kept cool and dry in storage. The four-digit UN identification number is 1449. The primary uses of barium peroxide are in bleaching, in thermal welding of aluminum, as an oxidizing agent, and in the dyeing of textiles.
Barium peroxide is a grayish-white powder.
BaO2 is an iron gray or white powder. It is slowly
decomposed in air, forming the hydroxide and oxygen.
It does not dissolve in water, but can slowly hydrolyze,
forming hydrogen peroxide in solution. Barium peroxide is a strong oxidizing agent and will explode if direct contact with organic matter occurs. Therefore, barium peroxide is always diluted to form a slurry before usage.
Barium peroxide is a strong oxidizing agent that is
used for bleaching. Barium peroxide contains O22-
subunits wherein the oxygen atoms bond to each other
as well as to the barium.
Bariumperoxide is used as a hydrogen peroxide source
and oxygen oxidant, as well as a bleaching agent. Its main
usage has been for making hydrogen peroxide and
oxygen, in organic syntheses, fabric printing and dyeing.
Barium peroxide is available commercially, primarily
as the oxctahydrate (which is the more stable form of
this peroxide).
Barium peroxide (BaO2) is a grayish-white dry powder that makes an excellent bleaching agent
that can be stored in paper packages. Its bleaching qualities are released when mixed with water.
Bleaching animal substances, vegetable fibers and straw; glass decolorizer; manufacture of H2O2 and oxygen; dyeing and printing textiles; with powdered aluminum in welding; in cathodes; in igniter compositions. Oxidizing agent in organic synthesis.
Barium peroxide is best prepared by reacting barium
nitrate with sodium peroxide in a cold solution:
Ba(NO3)2+Na2O2+xH2O→BaO2·xH2O+2NaNO3
The hydrated form is usually the octahydrate. If the
anhydrate is desired, the hydrated peroxide is dried
and then sintered at 350°C for 10 min or less:
4BaO2·xH2O+ heat→2BaO+2BaO2+xH2O+O2
About equal amounts of oxide and peroxide form.
The ratio is a function of the time and temperature of
heating. To separate the two forms, the heated mass is
plunged into a large volume of water where the
hydroxide is formed. The peroxide is insoluble whereas
the hydroxide is soluble, allowing the separation of the
two by filtration. The peroxide is then vacuum dried.
Barium peroxide, BaO2, was the first-known peroxo compound. It was used until mid-1900 in the manufacture of oxygen by the Brin process and of hydrogen peroxide by the Thenard reaction.
barium peroxide: A dense offwhitesolid, BaO2, prepared by carefullyheating barium oxide inoxygen; r.d. 4.96; m.p. 450°C. It is used as a bleaching agent. Withacids, hydrogen peroxide is formedand the reaction is used in the laboratorypreparation of hydrogen peroxide.
A grayish-white granular solid. Insoluble in water. Noncombustible, but accelerates the burning of combustible material. Mixture with finely divided combustible material may be explosive. Mixtures with combustible material may be ignited by friction or contact with moisture.
Decomposed by water. Insoluble in water.
Barium peroxide is a strong oxidizing agent. Contact with water can produce a temperature and oxygen concentration high enough to ignite organic materials [Bretherick's, 5th ed., 1995, p. 94]. Reacts explosively with acetic anhydride due to the formation of acetyl peroxide [Rust, 1948, p. 337]. Ignites when mixed with powdered aluminum, powdered magnesium or calcium-silicon alloys. Wood may ignite with friction from the peroxide. Decomposes when heated to 700°C to produce barium oxide and pure oxygen [Sax, 9th ed., 1996, p. 317]. Forms highly reactive mixtures with fuel-type materials.
Oxidizing material. Fire and explosion risk
in contact with organic materials. Keep cool and
dry. Toxic by ingestion, skin irritant.
Inhalation causes irritation of mucous membranes, throat, and nose. Contact with eyes or skin causes severe burns. Ingestion causes excessive salivation, vomiting, colic, diarrhea, convulsive tremors, slow, hard pulse, and elevated blood pressure; hemorrhages may occur in the stomach, intestines, and kidneys; muscular paralysis may follow.
Behavior in Fire: Can increase intensity of fire.
Flammability and Explosibility
Not classified
A poison via
subcutaneous route. A powerful oxidtzer.
Explodes on contact with acetic anhydride.
Ignites when mixed with calcium-silicon
alloys, powdered aluminum, powdered
magnesium, water + organic compounds.
Mixtures with propane react violently when
heated. The powder ignites when heated to
265℃ with selenium. Wood ignites with
friction from the peroxide. Incompatible
with H2S, water, peroxyformic acid,
hydroxylamine solution, mixture of (Mg +
Zn + Ba(NO3)2), and organic matter. See
also BARIUM COMPOUNDS (soluble)
and PEROXIDES, INORGANIC.
Is used as a bleaching agent; in making
hydrogen peroxide, oxygen; in aluminum welding; in
textile dyeing and for bleaching fibers; animal substances.
If this chemical gets into the eyes, remove anycontact lenses at once and irrigate immediately for at least15 min, occasionally lifting upper and lower lids. Seek medical attention immediately. If this chemical contacts the skin,remove contaminated clothing and wash immediately withsoap and water. Seek medical attention immediately. If thischemical has been inhaled, remove from exposure, begin rescue breathing (using universal precautions, including resuscitation mask) if breathing has stopped and CPR if heart actionhas stopped. Transfer promptly to a medical facility. Whenthis chemical has been swallowed, get medical attention.Give large quantities of water and induce vomiting. Do notmake an unconscious person vomit.
Color Code—Yellow: Reactive Hazard; Store in alocation separate from other materials, especially flammables and combustibles. Store in tightly closed containersin a cool, well-ventilated area away from water or moisture,and away from contact with the incompatible materialscited above. See OSHA Standard 1910.104 and NFPA 43ACode for the Storage of Liquid and Solid Oxidizers fordetailed handling and storage regulations.
UN1449 Barium peroxide, Hazard Class: 5.1;
Labels: 5.1—Oxidizer, 6.1—Poisonous materials.
A strong oxidizer. Keep away from
organic and combustible materials (such as wood, paper,
oil, fuels, and other easily oxidized materials) and peroxyformic
acid, hydrogen sulfide and hydroxylamine solutions,
since violent reactions occur.
Dispose of contents and container
to an approved waste disposal plant. All federal,
state, and local environmental regulations must be
observed. Contact your local or federal environmental protection
agency for specific recommendations.