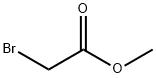
Methyl bromoacetate is used in the synthesis of coumarins and cis-cyclopropanes. It reacts with the conjugate base of (methylmethoxycarbene)pentacarbonylchromium(0) to prepare alkylated carbene complexes. Further, it is used to make vitamins and pharmaceuticals.
Methyl bromoacetate was used in the synthesis of novel coumarins. It was also employed in the synthesis of cis-cyclopropanes
Methyl Bromoacetate is an organic building block that has been used as a reactant in the preparation of isoquinolinone indole acetic acid derivatives as antagonists of chemoattractant receptor homologous molecule expressed on Th2 cells (CRTH2) for the treatment of allergic inflammatory diseases.
A colorless to straw-colored liquid with a sharp penetrating odor. Denser than water and soluble in water. Severly irritates skin and eyes. Toxic by ingestion and inhalation. Used to make vitamins and pharmaceuticals.
Highly flammable. Soluble in water.
Methyl bromoacetate is a halogenated ester. Esters react with acids to liberate heat along with alcohols and acids. Strong oxidizing acids may cause a vigorous reaction that is sufficiently exothermic to ignite the reaction products. Heat is also generated by the interaction of esters with caustic solutions. Flammable hydrogen is generated by mixing esters with alkali metals and hydrides.
Vapor is strong irritant to eyes.
TOXIC; inhalation, ingestion or contact (skin, eyes) with vapors, dusts or substance may cause severe injury, burns or death. Bromoacetates and chloroacetates are extremely irritating/lachrymators. Reaction with water or moist air will release toxic, corrosive or flammable gases. Reaction with water may generate much heat that will increase the concentration of fumes in the air. Fire will produce irritating, corrosive and/or toxic gases. Runoff from fire control or dilution water may be corrosive and/or toxic and cause pollution.
HIGHLY FLAMMABLE: Will be easily ignited by heat, sparks or flames. Vapors form explosive mixtures with air: indoors, outdoors and sewers explosion hazards. Most vapors are heavier than air. They will spread along ground and collect in low or confined areas (sewers, basements, tanks). Vapors may travel to source of ignition and flash back. Substance will react with water (some violently) releasing flammable, toxic or corrosive gases and runoff. Contact with metals may evolve flammable hydrogen gas. Containers may explode when heated or if contaminated with water.
Poison by intravenous
route. When heated to decomposition it
emits toxic fumes of Br-. See also ESTERS.