Misoprostol is a synthetic prostaglandin E1 analog that is commonly used for medical abortion, management of miscarriage, cervical priming, management of postpartum hemorrhage, and induction of labor. Because of misoprostol’s wide use in reproductive health, the drug is in the World Health Organization (WHO) Model List of Essential Medicines. As a synthetic analog of prostaglandin E1, misoprostol can avoid the major side effects caused by PGE1, including fetal bradycardia, pregnant women’s emesis, somnolence, and headache.
Routes for administering misoprostol include orally, sublingually, vaginally, rectally, or buccally. Pharmacokinetics studies carried out to compare vagina and oral administration have revealed that vaginal misoprostol is linked to slower clearance, lower peak plasma levels, and slower absorption, similar to an extended release preparation. Conversely, vaginal misoprostol is associated with more significant effects on the uterus and cervix due to the higher overall exposure to the drug.
Administration through the rectum revealed a similar pattern to vaginal administration; however, it has lower AUC and lower maximum peak concentration. The sublingual route has higher peak levels and more rapid absorption as compared to oral or vaginal administration.
Inducing Labor
Misoprostol is an effective and safe way of inducing labor although the Food and Drug Administration (FDA) has not yet approved it for use in pregnancy. However, the European Union (EU) approved its use after a study that was done between 2002 and 2012. It acts by causing uterine ripening and constrictions of the cervix.
Abortion
Misoprostol is normally used in combination with other drugs, such as methotrexate or mifepristone for medical abortion. It works by softening the cervix, causing it to open. Also, it makes the uterine muscles contract, thus inducing labor to expel the fetus. Misoprostol is a similar to prostaglandin which as a hormone that can cause a stronger reaction from the uterus. Most of the time, the drug is given in a clinic or hospital birthing centers.
Misoprostol can be best administered when one has passed 12 or more weeks of pregnancy to avoid possible severe complications. It can also be administered if there is a possibility of ectopic pregnancy or if one has an intrauterine device (IUD).
Misoprostol can be used either vaginally or orally if the pregnancy is still in the first trimester. The pregnant woman is advised to use 12 tablets of 200 grams and should put four tablets into the vagina or under the tongue and let it dissolve for 30 minutes. The woman is further advised to wait for 3 hours and repeat with four more pills in the vagina or under the tongue for 30 minutes.
The drug is more effective when inserted into the vagina; however, doctors usually administer it orally due to the possibility of infection when used vaginally. When used together with mifepristone, misoprostol is effective in approximately 95% of early pregnancies.
Postpartum Hemorrhage
Misoprostol is normally used both as treatment and prevention of postpartum hemorrhage secondary to its uterotonic properties. Studies have revealed that orally administered misoprostol is less effective as compared to oxytocin, but it still plays a vital role in treating postpartum hemorrhage when other agents fail or are not available.
Prevention of Ulcer
Misoprostol is commonly used for the treatment and prevention of NSAID-induced gastric and duodenal ulcers due to its analgesic and anti-inflammatory properties. It acts by inhibiting gastric acid inhibition of adenylate cyclase by G-protein coupled receptor that leads to decreased proton pump activity and decreased intracellular cyclic AMP levels at the apical surface of the parietal cell.
Failed Miscarriage
Misoprostol is used in treating a mother in case of fetal death that can result in miscarriage. It is also used in the termination of pregnancy for fetal anomalies. Initially, a low dose is administered and doubled for the extra doses until delivery.
The general adverse reactions experienced include contractions, abdominal pain, and GI intolerance and diarrhea. Many clinical studies revealed that about 13% of patients experienced diarrhea in the initial stages of therapy.
The most often reported side effects of orally taking the drug for the prevention and treatment of gastric ulcers include nausea, abdominal pain, headache, flatulence, dyspepsia, constipation, nausea, and constipation. Many patients experience fever when given multiple doses every 4 to 6 hours.
Women with wanted pregnancies are not supposed to take misoprostol to reduce the risk of increased uterine contractions and tone due to NSAID-induced gastric ulcers. This may cause complete or partial abortions or may lead to pregnancies with linked to congenital disabilities.
The drug may cause uterine hyperstimulation, which can affect the blood supply to the uterus negatively, thus increasing the chances of developing complications such as rupture of the womb.
There have been numerous controversies regarding the use of misoprostol for induction of labor due to various reasons. The use of misoprostol and mifepristone for medical abortion has the potential of improving access to abortion services; therefore, political opponents of abortion may view the drugs as a target and a threat. It is noteworthy that in 2000, the FDA approved the use of mifepristone and misoprostol in conjunction with misoprostol for the termination of early pregnancy, yet misoprostol was not approved for the same purpose.
Misoprostol is an orally-active PGEl analog useful in the treatment of gastric and
duodenal ulcers. As with other agents of this type, misoprostol may prove useful
as a gastric cytoprotective when administered concurrently with irritating drugs
such as nonsteroidal antiinflammatories.
Misoprostol is a yellow, viscous liquid with a musty odour. Miscible with ethanol, ether or chloroform, extremely insoluble in water or n-hexane. Very unstable at room temperature and undergoes thermal epimerization to the 8-isomer. It is also extremely sensitive to the Ph value. It removes the hydroxyl group at the 11α position in acidity and alkalinity, converts it into A-type prostaglandins, and isomerizes into B-type prostaglandins. However, it is relatively stable in the dispersion system of hydroxypropyl methylcellulose and can be stored at room temperature.
Misoprostol is a cytoprotective prostaglandin PGE1 analogue that inhibits gastric acid secretion. It has many medical uses, including nine obstetric indications. The drug was originally produced by Pfizer for the treatment of gastric ulcers. Misoprostol is commonly used to prevent NSAID-induced gastric ulcers in patients at high risk of complications from a gastric ulcer, such as elderly patients and patients with a history of ulcer. It has also been used in treating duodenal ulcers unresponsive to histamine H2-antagonists; the drug does not prevent duodenal ulcers,however, in patients taking NSAIDS. Misoprostol can cause miscarriage, often associated with potentially dangerous bleeding.
synthesis of Misoprostol
To a 1000 ml dried flask under a nitrogen atmosphere was added 74.6 g of(E)-trimethyl-[[1-methyl-1-[3-(tributylstannyl)-2-propenyl]pentyl]oxy]silane,125 ml anhydrous THF and 24.2 g of copper (I) iodide. The mixture wasstirred at room temperature for 30 minutes and then it was cooled to -25 to -30°C. 98.8 ml of methyllithium (2.86 M) in DEM was added dropwise and theresultant solution was stirred at -15°C for 2 hours. Then the reaction mixturewas cooled to -78°C and 25 g of methyl-5-oxo-3-[(triethylsilyl)oxy]-1-cyclopentene-1-heptanoate in 100 ml of THF was added rapidly. After stirringthe mixture for 5 min at -78°C, it was quenched into a mixture of 750 ml ofaqueous ammonium chloride solution and 200 ml of ammonium hydroxide.The resulting mixture was warmed to room temperature and stirred until adeep blue aqueous layer was obtained. Ethyl acetate (250 ml) was used forextraction. Then the combined organic layers were washed with brine andsubsequently dried over magnesium sulfate. After a filtration andconcentration under reduced pressure, an oil (105 g) was obtained. This oilcontaining the protected prostaglandin was subjected to acidic deprotection(cat. PPTS, acetone and water) and purification (chromatography on silica gel)to provide 15.8 g (60%) of misoprostol was identical.
Misoprostol (Cytotec), which is an analogue of
prostaglandin E1, has been approved for use in the prevention
of nonsteroidal antiinflammatory drug–induced
ulceration. It also is approved in other countries for the
treatment of peptic ulcer disease.
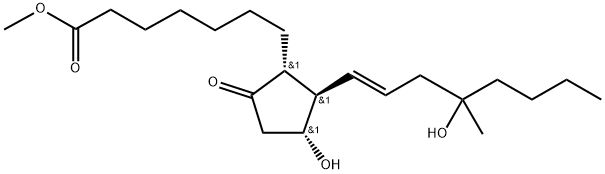
ChEBI: 7-[(1R,2R,3R)-3-hydroxy-2-(4-hydroxy-4-methyloct-1-enyl)-5-oxocyclopentyl]heptanoic acid methyl ester is a prostanoid.
Cytotec (Searle);Cyprostol;Oxaprost;Prostalgin.
Misoprostol, (16-(R,S)-methyl-16-hydroxy)-PGE1, methyl ester (Cytotec), is a modified prostaglandinanalog that shows potent gastric antisecretory and gastroprotectiveeffects when administered orally. Misoprostol isadministered orally in tablet form in a dose of 100 to 200 μg4 times a day to prevent gastric ulceration in susceptibleindividuals who are taking NSAIDs. Misoprostol is combinedwith the NSAID diclofenac (Voltaren) in an analgesic product(Arthrotec by Pharmacia), which is potentially safe for longtermantiarthritic therapy. This prostaglandin derivative absolutelyshould be avoided by pregnant women because of itspotential to induce abortion. In fact, the combined use of intramuscularmethotrexate and intravaginal misoprostol has beenclaimed to be a safe and effective, noninvasive method for thetermination of early pregnancy.
Cytoprotective prostaglandin E 1 analog that displays agonist activity at EP receptors. K i values are 120, 250, 67 and 67 nM at cloned mouse EP 1 , EP 2 , EP 3 and EP 4 receptors respectively. Prevents NSAID-induced gastric ulceration.
Misoprostol is absorbed
rapidly after oral administration and is hydrolyzed
to the active compound. It is metabolized by
the liver and excreted mainly in the urine. Adverse effects
include crampy abdominal pain, dose-related diarrhea,
and uterine contractions. The last-named effect
has led to its use in the control of postpartum bleeding.
Benign gastric and duodenal ulceration and NSAID
associated ulceration
Prophylaxis of NSAID induced ulceration
Veterinary Drugs and Treatments
Misoprostol may be useful as primary or adjunctive therapy in
treating or preventing
gastric ulceration, especially when caused
or aggravated by non-steroidal antiinflammatory drugs (NSAIDs).
Misoprostol is most useful to prevent GI ulceration or GI adverse
effects (anorexia, vomiting) associated with NSAID therapy. While
it can be used for treating gastric ulcers, other drugs are probably
just as effective and less expensive. It does not appear to be very
effective in reducing gastric ulceration secondary to high dose corticosteroid
therapy
Misoprostol may be efficacious in reducing or reversing cyclosporine-
induced nephrotoxicity. More data is needed to confirm
this effect.
One study demonstrated that misoprostol can reduce the clinical
signs associated with atopy somewhat in dogs.
Misoprostol’s effects on uterine contractibility and cervical softening/
opening make it effective as an adjunctive treatment in pregnancy
termination.
Rapidly metabolised to its active form (misoprostol acid)
after oral doses. Misoprostol acid is further metabolised
by oxidation in several body organs and is excreted mainly
in the urine.
Misoprostol is a synthetic prostaglandin E1 analogue which has ulcer healing, gastric acid antisecretory and mucosal protective properties. The antisecretory activity is mediated by direct action on specific prostaglandin receptors on the surface of gastric parietal cells. In dogs with innervated Pavlov pouches, inhibition of secretion is achieved at a lower dosage by intrapouch injection than by intravenous or intragastric administration, suggesting that the local effect may predominate. The mucosal protective effect against various damaging agents has been demonstrated in humans with doses that inhibit and doses which minimally affect acid secretion.