Propionic anhydride is a colorless liquid. Strong, pungent, unpleasant odor. Soluble in methanol, ethanol, ether, chloroform and alkali, decomposes in water.
Propionic anhydride is used as a esterifying agent for certain perfume oils, fats, oils, and especially cellulose and as a reagent in organic synthesis. In the production of alkyd resins, dyestuffs and drugs. Has been used as a dehydrating agent in some sulfonations and nitrations.
Propionic anhydride was previously used in the preparation of α and β-1-propionyl derivatives of glucopyranose tetra-acetate.
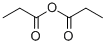
Propionic anhydride is an organic compound with the molecular formula (CH3CH2CO)2O. it is an colorless acid anhydride that is widely used as a reagent in organic synthesis.
Decomposes exothermically in water to form a corrosive solution of propionic acid [Merck, 11th ed. 1989].
Propionic anhydride reacts exothermically with water. The reactions are sometimes slow, but can become violent when local heating accelerates their rate. Acids accelerate the reaction with water. Incompatible with acids, strong oxidizing agents, alcohols, amines, and bases.
Strong irritant to tissue.
Inhalation causes irritation of eyes and respiratory tract. Contact with liquid causes burns of eyes and skin. Ingestion causes burns of mouth and stomach.
Combustible material: may burn but does not ignite readily. Substance will react with water (some violently) releasing flammable, toxic or corrosive gases and runoff. When heated, vapors may form explosive mixtures with air: indoors, outdoors and sewers explosion hazards. Most vapors are heavier than air. They will spread along ground and collect in low or confined areas (sewers, basements, tanks). Vapors may travel to source of ignition and flash back. Contact with metals may evolve flammable hydrogen gas. Containers may explode when heated or if contaminated with water.
Flammability and Explosibility
Non flammable
Moderately toxic by
ingestion. Mildly toxic by skin contact. A
corrosive irritant to skin, eyes, and mucous
membranes. Combustible when exposed to
heat or flame; can react with oxidizing
materials. To fight fire, use CO2, dry
chemical. When heated to decomposition it
emits acrid smoke and irritating fumes. Used
as an esterifyng agent and dehydrating
agent. See also ANHYDRIDES.
Propanoic anhydride is strong smelling and corrosive, and will cause burns on contact with skin. Vapour can burn eyes and lungs.
Propanoic anhydride has been prepared by dehydration of propanoic acid using ketene :
2 CH3CH2CO2H + CH2= C= O → (CH3CH2CO)2O + CH3CO2H.
Used in the manufacture of perfumes,
flavorings, alkyd resins; dyestuffs, pharmaceuticals; as an
esterifying agent for fats, oils, and cellulose; dehydrating
medium for nitrations and sulfonations.
UN2496 Propionic anhydride, Hazard class: 8;
Labels: 8-Corrosive material.
Shake the anhydride with P2O5 for several minutes, then distil. [Beilstein 2 IV 722.]
Vapors may form explosive mixture with
air. Incompatible with oxidizers (chlorates, nitrates, perox-
ides, permanganates, perchlorates, chlorine, bromine, fluo-
rine, etc.); contact may cause fires or explosions. Keep away from alkaline materials, strong bases, strong acids,
oxoacids, epoxides, reducing agents; alcohols and metals.
Contact with water forms heat 1 flammable propionic acid.
Compounds of the carboxyl group react with all bases, both
inorganic and organic (i.e., amines) releasing substantial
heat, water and a salt that may be harmful. Incompatible
with arsenic compounds (releases hydrogen cyanide gas),
diazo compounds, dithiocarbamates, isocyanates, mercap-
tans, nitrides, and sulfides (releasing heat, toxic and possibly
flammable gases), thiosulfates and dithionites (releasing
hydrogen sulfate and oxides of sulfur).
Use a licensed professional
waste disposal service to dispose of this material. Dissolve
or mix the material with a combustible solvent and burn
in a chemical incinerator equipped with an afterburner
and scrubber. All federal, state, and local environmental
regulations must be observed.