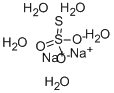
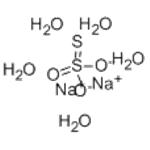
Sodium thiosulfate occurs as odorless and colorless crystals, a
crystalline powder or granules. It is efflorescent in dry air and
deliquescent in moist air.
translucent crystals or white powder
Sodium thiosulfate pentahydrate is used as an analytical reagent in laboratories. It is used in photographic processing and in gold extraction. Its alkylated derivatives, S-alkylthiosulfonates, are known as Bunte salts. It is involved in the preparation of thioglycolic acid. Furthermore, it is used to dechlorinate tap water, for removing free bromine from solutions and to check the pH of bleach solutions with liquid indicators. Other uses include tanning of leather; as an active component in hand warmers and chemical heating pads and as an anionic surfactant to aid in the dispersion of pharmaceutical preparations.
On an industrial scale, sodium thiosulfate is produced chiefly from
liquid waste products of sodium sulfide or sulfur dye manufacture.
Small-scale synthesis is done by boiling an aqueous solution of
sodium sulfite with sulfur.
ChEBI: Sodium thiosulfate pentahydrate is a hydrate consisting of sodium thiosulfate with 5 mol eq. of water. It has a role as an antidote to cyanide poisoning, a nephroprotective agent and an antifungal drug. It contains a sodium thiosulfate.
Sulfactol (Sterling Winthrop).
Pharmaceutical Applications
Sodium thiosulfate is used as an antioxidant in pharmaceuticals
(ophthalmic, intravenous, and oral preparations). It has also been
used for its antifungal properties and as a reagent in analytical
chemistry.
Sodium thiosulfate is used in ophthalmic, intravenous, and oral
pharmaceutical preparations. Apart from osmotic disturbances,
sodium thiosulfate is relatively nontoxic. It is moderately toxic by the subcutaneous route and mildly irritating to respiratory tract and
skin. Large oral doses have a cathartic action.
LD50 (IP, mouse) 5.6 g/kg
LD50 (IV, mouse) 2.4 g/kg
Sodium thiosulfate decomposes on heating. The bulk powder
should be stored in a cool place, and the container should be kept
tightly closed in a dry and well-ventilated place. It should not be
stored near acids.
Crystallise it from EtOH/H2O solutions or from water (0.3mL/g) below 60o by cooling to 0o, and dry it at 35o over P2O5 under vacuum. [Foerster & Mommsen Chem Ber 57 258 1924.] This salt is used as a secondary standard in volumetric analysis [Kilpatrick J Am Chem Soc 45 2132 1923], and is used as “Hypo” in photography [Hargreaves & Dunningham J Soc Chem Ind 42 147T 1923.]
Sodium thiosulfate is incompatible with iodine, with acids, and with
lead, mercury, and silver salts. It may reduce the activity of some
preservatives, including bronopol, phenylmercuric salts, and
thimerosal.
GRAS listed. Included in the FDA Inactive Ingredients Database (IV
solutions; ophthalmic solutions and suspensions; oral capsules,
solutions, and tablets). Included in the Canadian List of Acceptable
Non-medicinal Ingredients.