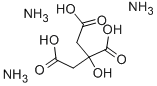
Ammonium citrate (various forms) is a white
powdery material with a slight ammonia-like odor.
Ammonium citrate tribasic is mainly used for chemical analysis, in industry for water treatment, in food industry for buffering agent and emulsifier, for metal surface cleaning (in particular petroleum pipeline cleaning), as a ceramic dispersant,as auxiliary infiltration agent, as detergent material and as soil improvement agent component.Ammonium citrate tribasic is applied in electroplating industry for non cyanide plating complexing agent, used mostly for preparing anti rust agent in machinery industry and as buffering agent. It serves as a chemical reagent in analytical chemistry to determine the efficient phosphate levels in phosphate salts and fertilizer.
Ammonium citrate tribasic is widely utilized as a buffering agent and emulsifier in food industry. It acts as a complexing agent in electroplating industry and is also used for metal surface cleaning. It is employed in chemical analysis and in water treatment. It serves as a chemical reagent in analytical chemistry to determine the efficient phosphate levels in phosphate salts and fertilizer. It is used in industries for water treatment, as a ceramic dispersant, a detergent material and a component for soil improvement. It is used in medical and electronics industries as an auxiliary infiltration agent.
ChEBI: A citrate salt in which all three carboxy groups are deprotonated and associated with ammonium ions as counter-cations.
Ammonium citrate is a white, granular, water-soluble
salt of ammonia and citric acid. It is used in a neutral
solution for determining the phosphorus content of
phosphate fertilizers (such as calcium hydrogen
phosphate (CaHPO4)), that are insoluble in water but
dissolve in complexing citrate solutions.
The fertilizer residue, after extraction of watersoluble
phosphorus, is re-extracted with a neutral or
strongly alkaline solution of 1 normal (1N) ammonium
citrate for a prescribed period, and the slurry filtered.
Citrate-soluble phosphorus is the phosphorus content in
the filtrate and is expressed as a percentage by weight of
the fertilizer.
The extraction of phosphate with 1N ammonium
citrate is carried out routinely for 30 minutes, first at
room temperature and then at 4O℃ For neutral
ammonium citrate solution, a temperature of 65℃ is
preferred.
Some European countries use an alkaline ammonium
citrate solution, which extracts smaller amounts of
phosphorus. Some experts consider it to be better
correlated with phosphorus uptake and plant growth.
However, most countries use a neutral normal
ammonium citrate solution for extracting citrate soluble
phosphorus.
It is used to make pharmaceuticals;
rust-proofing compounds and plasticizers; as a food additive;
stabilizing agent.
UN3077 Environmentally hazardous substances,
solid, n.o.s., Hazard class: 9; Labels: 9-Miscellaneous hazardous
material, Technical Name Required.
Contact with strong caustics causes the
release of ammonia gas.
May be flushed to sewer with
large volumes of water.