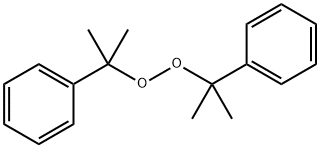
Dicumyl peroxide is a crystalline solid that melts at 42°C. It is
insoluble in water and soluble in vegetable oil and organic
solvents . It is used as a high-temperature catalyst in
production of polystyrene plastics. The deflagration hazard
potential of this peroxide was tested using 5 g of igniter in the
revised time–pressure test, but no pressure rise was produced
. Noller et al. found it to be an intermediate fire
hazard.
Dicumyl peroxide(DCP) is used:
in vulcanization of rubber
as a crosslinking agent in the synthesis of polylactic acid composite fibers
in the preparation of polyethylene composites
in the synthesis of polyamide 112/ethylene vinyl acetate copolymer blends.
Dicumyl peroxide is a pale yellow to white granular solid with a characteristic odor. It is used as a polymerization catalyst and vulcanizing agent.
The explosive instability of the lower dialkyl peroxides (e.g., dimethyl peroxide) and 1,1-bis-peroxides decreases rapidly with increasing chain length and degree of branching, the di-tert-alkyl derivatives being amongst the most stable class of peroxides. Though many 1,1-bis-peroxides have been reported, few have been purified because of the higher explosion hazards compared with the monofunctional peroxides. Dicumyl peroxide is unlikely that this derivative would be particularly unstable compared to other peroxides in it's class, Bretherick 2nd ed., p 44 1979.
Flammability and Explosibility
Non flammable
Mildly toxic by
ingestion. See also PEROXIDES. When
heated to decomposition it emits acrid
smoke and irritating fumes.
Crystallise the peroxide from 95% EtOH (charcoal). Store it at 0o. Potentially EXPLOSIVE. [Beilstein 6 IV 3220.]