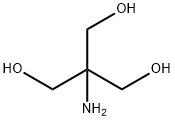
Tris, or tris(hydroxymethyl)aminomethane, or known during medical use as tromethamine or THAM, is an organic compound with the formula (HOCH2)3CNH2. It is extensively used in biochemistry and molecular biology as a component of buffer solutions such as in TAE and TBE buffers, especially for solutions of nucleic acids. It contains a primary amine and thus undergoes the reactions associated with typical amines, e.g. condensations with aldehydes. Tris also complexes with metal ions in solution. In medicine, tromethamine is occasionally used as a drug, given in intensive care for its properties as a buffer for the treatment of severe metabolic acidosis in specific circumstances. Some medications are formulated as the "tromethamine salt" including hemabate (carboprost as trometamol salt), and "ketorolac trometamol".
Tris(hydroxymethyl)aminomethane is a white crystal or powder. The melting point of Tris(hydroxymethyl)aminomethane is 171-172℃ and the boiling point of Tris is 219-220℃/1.3kPa. It is soluble in ethanol and water, slightly soluble in ethyl acetate and benzene, and insoluble in ether and carbon tetrachloride. It has a corrosive effect on copper and aluminum and has strong irritation.
Tris base, also known as THAM, is widely used as a biological buffer or as a component of buffer formulations such as TAE and TBE buffers. Tris has a pKa of 8.06 and is very useful in the biology and biochemistry lab because it has buffering capabilities in the range of the typical physiological pH of most living organisms (pH 7.0-9.0). Tris has been reported to interfere with the activity of a number of enzymes, it should therefore be used carefully when studying proteins.
1, Tris buffer, only nucleic acids and proteins are widely used as solvents.
2, Tris is used for protein crystal growth under different pH conditions.
3, Tris buffer having a low ionic strength can be used nematode (C. elegans) is formed lamin (lamin) intermediate fibers.
4, Tris is a major component of protein electrophoresis buffer.
5, Tris for the preparation of a surfactant, a vulcanization accelerator, and some drug intermediates.
6, Tris titrated also used as standard.
White, crystalline solid. PH (0.1M aqueous solution)
10.6, corrosive to copper, brass, aluminum.
Solubility in water 80 g/100 cc (20C). Combustible.
Trisaminol,Bellon,France,1964
tromethamine can help mask odor in a formulation. It is also used as a buffer, keeping product pH stable.
Tris base has been used:
- In the preparation of TAE buffer for TAE agarose gel used to run PCR products by gel electrophoresis
- As a component of sample buffer for SDS-PAGE
- In the preparation of IEC (ion-exchange chromatography) buffer
Tris(hydroxymethyl)aminomethane could used in the synthesis of surface-active agents, vulcanization accelerators, pharmaceuticals. As emulsifying agent for cosmetic creams and lotions, mineral oil and paraffin wax emulsions, leather dressings, textile specialties, polishes, cleaning Compounds, so-called soluble oils. Absorbent for acidic gases. Biological buffer.
Tris(hydroxymethyl)aminomethane is mainly required for preparation of buffers at physiological range of 7.3 to 7.5. The prepared buffers are compatible with biological fluids. It is of importance to laboratories as a standard pH solution.
Tris(hydroxymethyl)aminomethane has been used as a buffer solution for lactate dehydrogenase assay, in situ hybridization procedure and protein extraction from cells.
Tris(hydroxymethyl)aminomethane is used as a buffer in biochemistry and molecular biology laboratories. It is used as a primary standard to standardize acid solutions for chemical analysis. It finds application in cell membranes to increase permeability. As an alternative to sodium bicarbonate, it used in the treatment of metabolic acidosis. It acts as a precursor for the preparation of polymers, oxazolones and oxazolidines.
Tris(hydroxymethyl)aminomethane is prepared industrially by the exhaustive condensation of nitromethane with formaldehyde under basic conditions (i.e. repeated Henry reactions) to produce the intermediate (HOCH2)3CNO2, which is subsequently hydrogenated to give the final product.
ChEBI: Tris is a primary amino compound that is tert-butylamine in which one hydrogen attached to each methyl group is replaced by a hydroxy group. A compound widely used as a biological buffer substance in the pH range 7--9; pKa = 8.3 at 20 ℃; pKa = 7.82 at 37 ℃. It has a role as a buffer. It is a triol and a primary amino compound. It is a conjugate base of a member of Htris.
Nitromethane is reacted with formaldehyde to give
tris(hydroxymethyl)nitromethane in an initial step. This intermediate may be
reduced by catalytic hydrogenation (US Patent 2,174,242) or by electrolytic
reduction (US Patent 2,485,982).
Tris(hydroxymethyl)aminomethane is an established basimetric standard and buffer used in biochemistry and molecular biology. It may be used by itself as a buffer or as a component of mixed buffer formulations, such as Tris-EDTA (TE) buffer, Tris-acetate-EDTA (TAE) buffer, Tris-borate-EDTA (TBE) buffer, etc. It is pure, essentially stable, relatively non-hygroscopic and has a high equivalent weight.
Trizma base, commonly known as tris(hydroxymethyl)aminomethane is widely used as a biological buffer or as a component of buffer formulations like- Tris-EDTA (TE) buffer, Tris magnesium buffer or Tris-acetate-EDTA (TAE) buffer. It enhances the membrane permeability of cell membranes.
Tris(hydroxymethyl)aminomethane can ordinarily be obtained in highly pure form suitable for use as an acidimetric standard. If only impure material is available, it should be crystallised from 20% EtOH, aqueous MeOH (m 171.1o) or isopropanol (m 172-173o). Dry it in a vacuum desiccator over P2O5 or CaCl2. Alternatively, it is dissolved in twice its weight of water at 55-60o, filtered, concentrated to half its volume and poured slowly, with stirring, into about twice its volume of EtOH. The crystals which separate on cooling to 3-4o are filtered off, washed with a little MeOH, air dried by suction, then finally ground and dried in a vacuum desiccator over P2O5. It has also been recrystallised from water, MeOH or aqueous MeOH, and vacuum dried at 80o for 2 days. [Beilstein 4 H 303, 4 III 857, 4 IV 1903.]