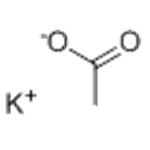
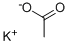Potassium acetate is the potassium salt of acetic acid. It is manufactured through the reaction between a potassium base such as potassium hydroxide or potassium carbonate with acetic acid. It is an important macromineral with many physiological functions and is required for nerve conduction, cardiac, skeletal and smooth muscle contraction, energy generation, nucleic acid synthesis as well as mainlining the blood pressure and normal renal function. It can be taken as a nutritional supplement with antihypertensive effects and prevention effects against hypokalemia. It has several other common applications: (1) as a deicer; (2) as a food additive; (3) molecular biology applications such as DNA purification, tissue preservation, fixation and mummification; (4) Death penalty (5) as the catalyst during the production of polyurethanes.
https://pubchem.ncbi.nlm.nih.gov/compound/Potassium_acetate#section=Top
https://en.wikipedia.org/wiki/Potassium_acetate
Potassium acetate (CH3CO2K) is the potassium salt of acetic acid.
Pure potassium acetate is odorless or has a faint acetic odor and a saline taste.
Potassium acetate, KC2H302, is a white, deliquescent solid, soluble in water and alcohol, insoluble in ether, that melts at 292°C. It is used as an analytical reagent, dehydrating agent, in medicine, and in crystal glass manufacture.
Potassium acetate is used in the manufacture of glass; as a softening agent for papers and textiles; as a dehydrating agent; and as a buffer. In medicine it is used as an expectorant and diuretic.
Potassium acetate was originally used in the preparation of Cadet's fuming liquid, the first organometallic compound produced. It is used as diuretic and urinary alkaliser, and acts by changing the physical properties of the body fluids and by functioning as an alkali after absortion.
1) Potassium acetate, crystalline powder, USP is, as its name suggests, the potassium salt of acetic acid and is used as a catalyst for producing polyurethanes2) In the food industry it is used as a food additive, an acidity regulator and a preservative
ChEBI: A potassium salt comprising equal numbers of potassium and acetate ions
Potassium acetate is used as a catalyst in the production of polyurethanes.
Potassium acetate can be used as a deicer instead of chloride salts such as calcium chloride or magnesium chloride. It offers the advantage of being less aggressive on soils and much less corrosive, and for this reason is preferred for airport runways. It is, however, more expensive. Potassium acetate is also the extinguishing agent used in class K fire extinguishers because of its ability to cool and form a crust over burning oils.
3 – 1 - Food additive
Potassium acetate is used as a food additive as a preservative and acidity regulator. In the European Union, it is labeled by the E number E261 ; it is also approved for usage in the USA and Australia and New Zealand. Potassium diacetate (CAS # 4251-29-0) with formula KH(O2CCH3)2 is a related food additive with the same E number as potassium acetate.
3 – 2 - Medicine and biochemistry
In medicine, potassium acetate is used as part of replacement protocols in the treatment of diabetic ketoacidosis because of its ability to break down into bicarbonate and help neutralize the acidotic state.
In molecular biology, potassium acetate is used to precipitate dodecyl sulfate (DS) and DS-bound proteins, allowing the removal of proteins from DNA. It is also used as a salt for the ethanol precipitation of DNA.
Potassium Acetate can be prepared by treating a potassium-containing base such as potassium hydroxide or potassium carbonate with acetic acid:
2 CH3COOH + K2CO3→ 2 CH3CO2K + CO2 + H2O
This sort of reaction is known as an acid-base neutralization reaction. Potassium acetate is the salt that forms along with water as acetic acid and potassium hydroxide are neutralized together.
Conditions/substances to avoid are: moisture, heat, flames, ignition sources, and strong oxidizing agents.
Potassium acetate is prepared by addition of potassium carbonate in a small volume of water to acetic acid solution, followed by evaporation and crystallization:
K2CO3+ 2CH3COOH →2CH3COOK + H2O
Potassium acetate is generally used as an acidity regulator, buffer, preservative and firming agent.
Flammability and Explosibility
Non flammable