Flucythrinate controls a wide range of insects on cotton, fruit trees,
vines, citrus, bananas, pineapples, olives, coffee, cocoa, hops, vegetables,
soyabeans, cereals, alfalfa, sugar beet, sunflowers and tobacco.
Flucythrinate belongs to the class of synthetic pyrethroids, primarily used to control Heliothis spp. in cotton and other insect pests in agricultural crops.
Insecticide, Acaricide: Not approved for use in EU countries. Not currently registered in the U.S. Flucythrinate is a synthetic pyrethroid used to control pests in apples, cabbage, head lettuce, pears, corn and cotton, but it was used primarily on cotton.
AASTAR®[C]; CYBOLT®; CYTHRIN®;
FUCHING JUJR®; GUARDIAN®[C]; PAYOFF®[C];
STOCK GUARD®; TOMAHAWK®
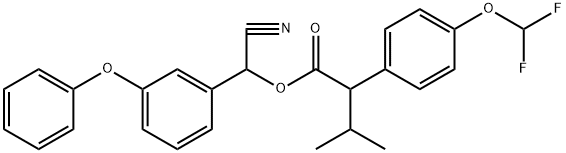
Flucythrinate
is the (S)-2-(4-difluoromethoxyphenyl)-3-methylbutyrate
analog of fenvalerate, partially resolved. Its insecticide
action is approximately two times higher compared
with fenvalerate (138,142).
Surface Water. The half-life of ?ucythrinate in an estuarine environment is 34 days (Schimmel et al., 1983).
Chemical/Physical. Hydrolyzes in aqueous solutions forming acetic acid and other compounds.
Flucythrinate is an analogue of fenvalerate and would be expected to
have a similar fate.
Flucythrinate is a stable compound but it is readily hydrolysed at alkaline
pH to afford 2-(4-difluoromethoxyphenyl)-3-methylbutyric acid (2),
3PBAl(3) and cyanide ion. Its DT50 values at 27 °C are 52 days (pH 5) and
6.3 days (pH 9). It undergoes aqueous and soil surface photolysis with
DT50 values of 1-4 days and <2 days, respectively. The products formed
under aqueous conditions were 2 and 3. Compound 3 was oxidised to
3PBA (4) and reduced to 3PBAlc (5). The α-carbamoyl (amide) derivative
(6) of 3PBAlc was also detected. On soil surfaces, products were similar
with the exceptions that 3PBA was not formed and the α-amide (7) and
a-carboxy derivative (8) were formed. These are usually considered to be
products of thermal degradation (Dureja and Chattopadhyay, 1995).