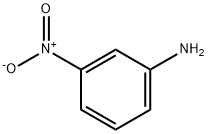
3-Nitroaniline is a ochre-yellow to orange crystalline powder, crystallizes as yellow needles from water. It is moderately soluble in organic solvents and sparingly soluble in water (0.11 %).
Yellow, rhombic crystals or powder. Finely dispersered particles form explosive mixtures.
Combustible.
Dyestuff intermediate. Acetylation of 3-nitroaniline followed by reduction gives 3-aminoacetanilide. Diazotization, followed by reduction of the diazosulfonate with ammonium bisulfite and subsequent hydrolysis, gives (3-nitrophenyl) hydrazine; an intermediate in the production of pyrazolone azo coupling components. 3-Nitroaniline is used as a diazo component (Fast Orange R Base) in azo dyes (e.g., C.I. Disperse Yellow 5 and C.I. Acid Orange 18).
3-Nitroaniline is commonly used as a raw material for dyes. It is also used as a chemical intermediate for azo coupling component 17 and the dyes disperse yellow 5 and acid blue 29. The chemical is changed to other substances (dyestuffs and m-nitrophenol) during the dyeing process.
1,3-Dinitrobenzene is added to warm water containing magnesium sulfate. An aqueous solution of sodium hydrogen sulfide (6 molar equivalents) is added gradually to the vigorously stirred emulsion, and reduction is completed by heating to 90 ℃. The 3-nitroaniline produced solidifies on cooling and is separated by filtration.
M-nitroaniline partial reduction.
Yellow needles or yellow powder.
Thermal stability of 3-Nitroaniline is reduced by various impurities. 3-Nitroaniline may be sensitive to prolonged exposure to light. 3-Nitroaniline may react explosively with ethylene oxide at 266° F. 3-Nitroaniline is incompatible with acids (nitric, sulfuric), acid chlorides, acid anhydrides, chloroformates and strong oxidizing agents. . Unstable when heated.
Moderate fire risk. Toxic when absorbed by
skin.
Flash point data for 3-Nitroaniline are not available; however, 3-Nitroaniline is probably combustible.
1,3-Dinitrobenzene is added towarmwater containing magnesium sulfate. An aqueous solution of sodium hydrogen sulfide (6molar equivalents) is added gradually to the vigorously stirred emulsion, and reduction is completed by heating to 90℃. The 3-nitroaniline produced solidifies on cooling and is separated by filtration.
Biological. A bacterial culture isolated from the Oconee River in North Georgia degraded 3-
nitroaniline to the intermediate 4-nitrocatechol (Paris and Wolfe, 1987). A Pseudomonas sp. strain
P6, isolated from a Matapeake silt loam, did not grow on 3-nitroaniline as the sole source of
carbon. However, in the presence of 4-nitroaniline, all of the applied 3-nitroaniline metabolized
completely to carbon dioxide (Zeyer and Kearney, 1983). In the presence of suspended natural
populations from unpolluted aquatic systems, the second-order microbial transformation rate
constant determined in the laboratory was reported to be 4.6 ± 0.1 x 10-13 L/organism?h (Steen,
1991).
In activated sludge inoculum, following a 20-d adaptation period, no degradation was observed
(Pitter, 1976).
Chemical/Physical. Reacts with acids forming water soluble salts.
Purify it as for o-nitroaniline. Warning: it is absorbed through the skin. [Beilstein 12 IV 1589.]