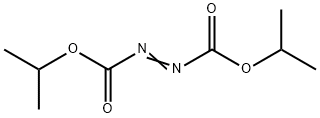
Isopropyl azodicarboxylate (Acronym DIAD; foaming agent DIPA) belongs to diisopropyl azo-hydroxy acid salt. At room temperature, it is an orange-red transparent oily liquid with special smell and is soluble in almost all organic solvents and plasticizers. It is also insoluble in water but soluble in common plasticizer. It can be well miscible with plastic with good thermal stability. Storage stability: the decomposition substance is colorless, non-toxic, non-pollution, non-blooming and odorless. In the range of 40~120 °C, it can obtain high gas evolution. It is also the liquid blowing agent of vinyl resin and can be used to make light-colored vinyl foam. It has a uniform pore structure, with different formulations and processing conditions obtaining the foam of either closed pore or open pore. It can also be used as pharmaceutical intermediates and organic synthesis reagents.
The above information is edited by the chemicalbook of Dai Xiongfeng.
It can be used as dye intermediates, pharmaceutical intermediates, organic synthesis reagents, and the foaming agent of the rubber plastics liquid.
Isopropyl azodicarboxylate can be used for the liquid foaming agent of vinyl resin. It can be used to make light-colored vinyl foam. It has a uniform pore structure with different formulations and processing conditions giving foam of either closed pore or open pore. It can also be used as pharmaceutical intermediates, and organic synthesis reagents.
Diisopropyl azodicarboxylate is used as an important reagent in the production of many organic compounds. It is used in association with triphenyl phosphine in Mitsunobu reaction of alcohols, acids and amides. It acts as reactant in the preparation of chromenes resembling classical cannabinoids, norbornene-based guanidine-rich polymers and acceptor-donor-acceptor organic dyes.
Diisopropyl Azodicarboxylate is an azodicarboxylic acid derivative used as a reagent in the production of many organic compounds. Diisopropyl Azodicarboxylate is commonly used as an oxidizer in Mitsun
obu reactions and can also serve as a selective deprotectant of N-benzyl groups in the presence of other protecting groups.
Reacted with dioxaphosphorinanes and iron catalyst to generate rearranged phosphonium ylide products.
Purify the azo compound by distillation at as high a vacuum as possible. Since it is likely to explode, use an oil bath for heating the still, and all operations should be carried out behind an adequate shield. [Kauer Org Synth Coll Vol IV 412 1963, Beilstein 3 III 233]. This reagent is useful in the Mitsunobu reaction [Mitsunobu Synthesis 1 1981, Gennari et al. J Am Chem Soc 108 6394 1986, Evans et al. J Am Chem Soc 108 6394 1986, Hughes Org React 42 335 1992, Dodge et al. Org Synth 73 110 1996, Hughes Org Prep Proc Int 28 127 1996, Ferguson & Marcelle J Am Chem Soc 128 4576 2006; see also di-tert-butyl azodicarboxylate and DEAD above].