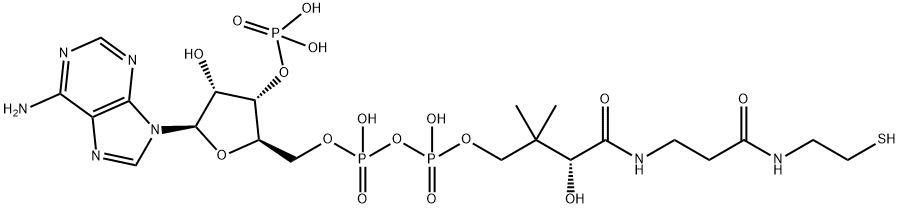
Coenzyme A (CoA) is an essential cofactor functioning as an acyl group carrier and carbonyl-activating group for the citric acid cycle and fatty acid metabolism. About 4% of cellular enzymes utilize CoA as a substrate. It is synthesized from pantothenic acid in a 5-step process that requires ATP. The pantothenate kinase step of the CoA biosynthetic pathway has been identified as a target for the development of antibacterial compounds.
Coenzyme A hydrate has been used in the thiolase enzyme assay of recombinant acetoacetyl-CoA thiolase (rACAT) in Clonorchis sinensis. It may be used as a reference standard in Raman spectra measurements.
A hydrated form of Coenzyme A (CoA) is a useful biochemical research chemical, used in the preparation of high-yielding cell-free protein synthesis platforms.
ChEBI: Tetraanion of coenzyme A.
Coenzyme A is structurally composed of 2-aminoethanethiol, pantothenic acid and phosphorylated adenosine phosphate.
Coenzyme A is a cofactor involved in acyl group transfer. It is essential for carbohydrate, lipid and protein metabolism. Coenzyme A acts as a cofactor for vitamin B coenzymes.
The white powder is best stored in an inert atmosphere in the dark in sealed ampoules after drying in vacuo over P2O5 at 34o. It has UV: max 259 nm (� 16,800) in H2O. [Buyske et al. J Am Chem Soc 76 3575 1954.] It is soluble in H2O but insoluble in EtOH, Et2O and M2CO. It is readily oxidised in air and is best kept as the more stable trilithium salt [Moffat & Khorana J Am Chem Soc 83 663 1961; see also Beinert et al. J Biol Chem 200 384 1953, De Vries et al. J Am Chem Soc 72 4838 1950, Gregory et al. J Am Chem Soc 74 854 1952 and Baddiley Adv Enzymol 16 1 1955]. [Beilstein 26 III/IV 3663.]