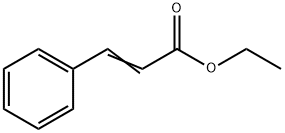
Ethyl cinnamate is the ester of cinnamic acid and ethanol, and is typically found in the essential oil of cinnamon. It is noteworthy that pure ethyl cinnamate has an aroma of cinnamon, complicated by balsamic and red fruit nuances,all of which can be characteristic of a fine Nebbiolo wine's aromatics.
Ethyl Cinnamate, ethyl 3-phenylacrylate, C6H5CH=CHCOOC2H5.Occurs naturally in storax and the volatile oil of Koempferia galanga.The commercial product is a colourless liquid, invariably prepared artificially. Has a sweet balsami honey-note odor of great persistence. Ethyl cinnamate is useful in Oriental bouquets, and in combination with clary sage and citrus oils will make delightful amber colognes.
Normally occurring in the trans-form; a cis-form also exists. Reported found in Oriental styrax, in the oil of
Campheria galanga and in the rhizomes of Hedychium spicatum. Also reported found in cherry, American cranberry, pineapple,
guava, strawberry, fresh blackberry, strawberry jam, soybean, yellow passion fruit juice, hybrid passion fruit juice, apple brandy,
quince, prickly pear, strawberry wine, Bourbon vanilla, sea buckthorn, cinnamon leaf and root bark, clove, brandy, rum, sherry,
grape wines, cocoa, soybean and other natural sources.
Ethyl cinnamate is used in medical intermediate, as a pharmaceutical intermediate and in organic synthesis.
Ethyl cinnamate is found in storax oil, Kaempferia galanga,
and several other oils. It is produced by the direct esterification esterification
of ethanol with cinnamic acid under azeotropic conditions
or by Claisen-type condensation of ethyl acetate and
benzaldeyde in the presence of sodium metal .
By heating to 100°C cinnamic acid, alcohol and sulfuric acid in the presence of aluminum sulfate; also by Claisen con�densation of benzaldehyde and ethyl acetate
ChEBI: Ethyl cinnamate is an alkyl cinnamate and an ethyl ester.
Taste characteristics at 20 ppm: balsamic, powdery, fruity, berry, punch, spice, sweet and green.
Ethyl cinnamate can be used as a flavoring and fragrance ingredient. It is one of the key odor components reported in Burgundy Pinot noir wines.
Ethyl cinnamate inhibits the growth of Chlorella pyrenoidosa.
Moderately toxic by
ingestion. Combustible liquid. When heated
to decomposition it emits acrid smoke and
irritating fumes. See also ESTERS
Wash the ester with aqueous 10% Na2CO3, then water, dry (MgSO4), and distil it. The purified ester is saponified with aqueous KOH, and, after acidifying the solution, cinnamic acid is isolated, washed and dried. The ester is reformed by refluxing for 15hours the cinnamic acid (25g) with absolute EtOH (23g), conc H2SO4 (4g) and dry *benzene (100mL), after which it is isolated, washed, dried and distilled under reduced pressure [Jeffery & Vogel J Chem Soc 658 1958]. [Beilstein 9 IV 2006.]