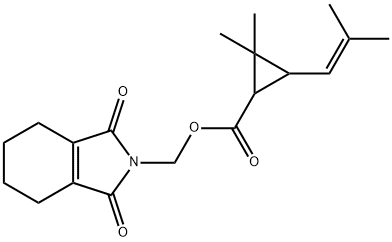
Form: Colorless crystals with slight pyrethrum-like odor
Tetramethrin is a synthetic pyrethroid pesticide used in large-scale commercial agricultural applications as well as in consumer products for domestic purposes.
Tetramethrin is used to control flies, wasps, cockroaches and other
insects in public health, home and garden situations.
ChEBI: Tetramethrin is a phthalimide insecticide, a member of maleimides and a cyclopropanecarboxylate ester. It has a role as a pyrethroid ester insecticide. It is functionally related to a chrysanthemic acid.
Colorless crystals with slight odor. Non corrosive. Used as an insecticide.
Hydrolysis occurs with strong acid or base.
A pyrethroid. Tetramethrin is an ester and nitrile. Esters react with acids to liberate heat along with alcohols and acids. Strong oxidizing acids may cause a vigorous reaction that is sufficiently exothermic to ignite the reaction products. Heat is also generated by the interaction of esters with caustic solutions. Flammable hydrogen is generated by mixing esters with alkali metals and hydrides.
Insecticide: Tetramethrin is often formulated as an aerosol and
used primarily for indoor pest control or in mosquito coils.
It is also used in shampoos to control fleas and ticks on
pets. It is often formulated with other insecticides and
synergists. Not approved for use in EU countries.
Registered for use in the U.S.
FMC 9260®; ENT-27339; EVERCIDE
INTERMEDIATE® 2265 (tetramethrin + fenvalerate);
MULTICIDE®; NEO-PYNAMIN®; NEOPYNAMINE®;
NEOPYNAMIN FORTE®; NIAGARA®-9260; NIA®-
9260; PHTHALTHRIN®; SP-1103; SUMITOMO®
SP-1103
Tetramethrin was the second synthetic pyrethroid to be produced commercially
(1964). The compound has a rapid knock-down action and a
reasonable killing activity which is enhanced by use with synergists. It is
usually used with piperonyl butoxide and in the presence of other insecticides.
It has two chiral centres, both in the acid group, and therefore is a
1 RS-cis-trans mixture. A(1R)-rich product containing cis- and trans-isomers
in the ratio 20:80 is also used in public health; this is named dtetramethrin.
The two products will not be distinguished here because
metabolic studies have been performed with various combinations of
isomers. Most published information relates to its mode of action in
insects and its metabolism in rodents. This is a reflection of its limited
outdoor and field use.
Acute oral LD50 for rats: >5,000 mg/kg
Tetramethrin is a stable chemical but it is base labile and it is also sensitive
to strong acids. It is hydrolysed to (1RS)-cis-trans-2,2-dimethyl-3-(2-
methylprop-1-enyl)cyclopropanecarboxylic acid (chrysanthemic acid, 2)
and tetrahydrophthalimide (4). It is oxidised by m-chloroperbenzoic acid
to form epoxy-tetramethrin (5) which is ring-opened to the diol (6) in
dilute aqueous acid (Smith and Casida, 1981). This reaction has been
postulated as initiating the opening of the cyclopropane ring with the
ultimate formation of CO 2 in biological systems (see below). Another
biomimetic reaction of tetramethrin and its ester cleavage product 4 is
Michael addition of thiols to the double bond. Glutathionyl-tetramethrin
(7) is formed by incubation of the constituents in buffered aqueous
methanol (Smith et al., 1982). Its formation on incubation with mouse liver
microsomes is probably non-enzymatic. At the time of this discovery
there appeared to be no equivalent reaction in vivo but the more recent
discovery of sulfonate metabolites (see below) demonstrates its role.
The chrysanthemic acid esters are very sensitive to photodegradation,
being subject to ring-opening initiated by the formation of epoxides such
as 5 (Ruzo et al., 1982) as described under bioallethrin and phenothrin.
Isomerisation was observed only in de-oxygenated benzene solution
because photo-oxidative degradation predominated under most conditions.
Many products were seen in oxygenated benzene solution and in a
thin film in sunlight.
These chemical and photochemical reactions are summarised in
Scheme 1.