Flutolanil is a fungicide that has been used for controlling brown patch on creeping bentgrass fairways.
Flutolanil is used to control Basidiomycetes diseases in rice,
cereals, sugar beet and other crops.
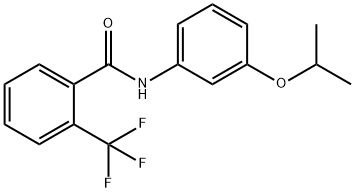
ChEBI: Flutolanil is a member of the class of benzamides, obtained by formal condensation of the carboxy group of 2-(trifluoromethyl)benzoic acid with the amino group of 3-(ispropyloxy)aniline. A fungicide used to control a range of pathogens especially Rhizoctonia spp. on rice, turf and other crops. It has a role as an EC 1.3.5.1 [succinate dehydrogenase (quinone)] inhibitor and an antifungal agrochemical. It is a member of benzamides, an aromatic ether, a member of (trifluoromethyl)benzenes and a benzanilide fungicide.
Low toxicity by ingestion, skincontact, intraperitoneal, and subcutaneous routes. Whenheated to decomposition it emits toxic vapors of NOx andF??.
Flutolanil is an analogue of mepronil in which the methyl group
is replaced by trifluoromethyl. Both compounds have systemic activity.
This change in structure should render flutolanil more biostable by
hindering hydrolysis and removing the option of methyl hydroxylation
and further oxidation. This seems to be borne out in practice in that
most of the metabolism of flutolanil occurs via O-dealkylation and aryl
hydroxylation. Hydrolysis has not been detected.
Flutolanil is a stable arylamide with no particularly weak link in its comparatively
simple chemistry. It is stable over the pH range 3-11 and it is
stable to heat (PM). It is stable in sunlight (PM) but it was slowly
degraded in 50% aqueous ethanol solution irradiated with a high pressure
mercury lamp whilst bubbling oxygen through the solution (Tsao and Eto,
1991). The study was conducted using non-radiolabelled compound. No
degradation occurred in the absence of oxygen. Even under these conditions,
the addition of photosensitisers was required to give a reasonable amount of breakdown. With 5% acetone in the solution, 20% degradation
was obtained in 8 hours. Almost no decomposition occurred on a glass
surface in 8 hours unless a sensitiser (e.g. benzophenone) was added. This
gave 40% decomposition.
The products in solution and on surfaces were different, as shown in
Scheme 1. The major product (80%) in solution was 2-(trifluoromethyl)-
benzamide (2). The benzoic acid (3) was identified as a minor product.
The N-ethoxycarbonyl derivative (4) was due to reaction with the solvent.
Amide bond cleavage was postulated to occur via oxidation in the aniline
ring (Tsao and Eto, 1991; Yumita et al., 1984). The resulting phenolic
products and anilines were converted into unidentified polar polymers.
No product 2 was obtained by irradiation on a glass surface (Tsao and
Eto, 1991). Under these conditions 3'-hydroxy-2-(trifluoromethyl)benzanilide
(5), i.e. dealkylated flutolanil, and a rearrangement product (6)
were the main products. Flutolanil is therefore an extremely stable
compound which undergoes slow photo-oxidation rather than aqueous
photolysis.