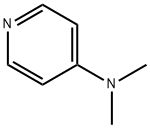
4-Dimethylaminopyridine(1122-58-3) is highly powerful catalyst of organic synthesis. The treatment of substrates such as alcohols, phenols and amines with acetic anhydride (or acetyl chloride) in the presence of pyridine has provided a general acetylation method since the turn of the 20th century. However, this approach often proves to be unsatisfactory for the acetylation of deactivated substrates. It was not until the late 1960’s that certain 4-dialkylaminopyridines were found (independently by two research groups)
[1, 2] to be much superior to pyridine as catalysts for difficult acetylations or acylations, in general.
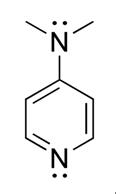
Figure 1 the chemical structure of DMAP
4-Dialkylaminopyridines were soon found to have general applicability for catalysis of a wide variety of reactions. 4-dimethylaminopyridine’s (DMAP) wide applicability has been frequently reviewed since the first review appeared in 1978. [3] The accelerating pace of reported applications for DMAP and the availability of DMAP in commercial quantities, at modest prices, have continued to stimulate great interest in its use as a catalyst in the fields of organic, polymer, analytical and biochemistry. Today there are thousands of examples of the use of DMAP in far ranging fields of chemistry in both patents and the research literature. Many full-scale production processes utilizing DMAP have been and are being operated. Several pharmaceutical and agricultural products that rely on DMAP’s superior catalytic properties in their synthetic sequences have been produced for years. Since 1976 more than 11,000 US patents have been granted which mention DMAP or dimethylaminopyridine.
The functional groups and class of compounds that are involved in the reactions with DMAP include alcohols, amines, arenes, azides, carbenes, enols, epoxides, hydrazines, hydroxylamines, phenols, thiols, lipids, sugars, aminoacids, peptides, alkaloids, steroids, terpenes, and others. Reactions that have been published in the literature using DMAP fall into, but are not limited to, the following types of reactions: Acylation; Acetylation; Alkylation; Benzoylation; Bischler-Naperalski cyclization, Carbonylation; Carbo-diimidation; Cyclization; Dehydration; Esterificaton; Indole Synthesis; Nucleophilic Substitution; Rearrangement; Silylation; Sulfonamidation; Sulfonation; Tritylation; Formylation; Carbamoylation; Phosphorylation; Lactonization; Pivaloylation; Dakin-West Reaction; Baylis-Hillman Reaction.
DMAP (m.p. 112-113°C,1122-58-3) and PPY (m.p. 57-58°C)[4] are colorless, crystalline substances which are very soluble in methanol, ethyl acetate, chloroform, methylene chloride, 1,2-dichloroethane, acetone, and acetic acid and less soluble in cold hexane, cyclohexane, and water. DMAP can be recrystallized from ethyl acetate and PPY from pentane or hexane. The basicities of DMAP[5] and PPY in water as well as the dipole moment of DMAP[7-9] in benzene and dioxane have been determined by several groups. Of especial interest are the thermodynamic investigations concerned with the protonation of DMAP in water[11] and calculations whereby the influence of substituents on the basicity has been determined[6, 9, 10].
DMAP reacts readily with electrophilic reagents. It is possible to quaternize DMAP in high yield with either methyl iodide or ethyl bromide, decomposes quantitatively in the presence of aqueous alkali to N-methy1-4-pyridone[17].
Addition of DMAP to S, S'-diethyl-S, S'-dimethyl-S, S-1, 2vinylenedisulfonium salts results in the smooth formation of the salt with concomitant generation of ethyl methyl sulfide[19]. Reaction of DMAP with acetylenedicarboxylic acid leads spontaneously to the bis-adduct in high yields[20].
On reaction with perbenzoic acid the strongly polar N-oxide is formed. Nitration of DMAP with HNO3/H2SO4 gives the 3-nitro derivatives in 81 % yield and, under forcing condition; the 3,5-dinitro compounds are obtained[16]. Reaction with O-(p-toluenesulfony1)hydroxylamine affords the N-amino compound in 67% yield which is isolated as the perchlorate. By treatment with D2O it is possible to selectively exchange the a-protons in DMAP, with DClO4 to exchange the P-protons to furnish and with D2O/NaOD to replace all aromatic protons by deuterium[18].
Application as acylation catalysts
Acylation of alcohol
The high catalytic activity of DMAP and PPY can be used for acylating sterically hindered secondary or tertiary alcohols with carboxylic anhydrides or acyl halides when the pyridine method fails. In most cases, it is necessary to use only 0.05-0.2 mol of catalyst per mol of substance and the acid that is formed can be bound with an equivalent amount of trimethylamine[21, 22] or pyridine[20]. Such solvents as hexane, toluene, benzene, methylene chloride, chloroform, ethyl acetate, tetrahydrofuran, triethylamine, pyridine, or acetic anhydride are suitable for use with these catalysts.
Among the tertiary alcohols which can be easily acylated with DMAP and PPY, mention should be made of l-methyl cyclohexanol, 1-ethynylcyclohexanol, 1,l-diphenylethanol, linalool, l, l-dimethoxy-2-methyl-3-buten-2-ol, 5,5-dimethoxy-2-methyI-3-pentyn-2-ol, and cis-4- (1-hydroxyisopropyl)-2-methylcyclohexanone.
Acylation of phenols
In the acylation of phenols, DMAP and PPY effect a similar increase in reaction rate as is found in the case of alcohols. Hence, the method is of interest for the acylation of sterically hindered phenols. For example, mesitol can be smoothly acetylated with acetic anhydride/DMAP to 2,5-ditert-butylphenol and analogous compounds can be transformed into acyl derivatives of the type in high yields[24]. 11,12-Dihydroglaziovine smoothly affords the acyl derivative[23, 25].
Acylation of amines
DMAP and PPY have been seldom used for the acylation of amines. The kinetic investigations of Lituinenko and Kirichenko [26] have shown that an enormous increase in reaction rate is observed when acylations are carried out in aprotic solvents. These authors have determined the following relative rate constants (in parentheses) for the amine-catalyzed acylation of m-chloroaniline with benzoyl chloride in benzene: N, N-dimethylaniline (0.1); triethylamine (0.072); 2,6-dimethylpyridine (0.03); pyridine (1.80); 4-methylpyridine (10.0); and DMAP (10600).
Acylation of enolates
Acylations involving CH-acid compounds which can be performed with pyridine or triethylamine as catalyst are found to proceed at a much higher rate when DMAP or PPY is used. The Dakin-West reaction of N-acyl amino acids, in which a 2-oxazolin-5-one is acetylated at C-4 with a carboxylic anhydride in pyridine with formation of a new C-C bond, has been extensively investigated[27]. The combination products, consisting of the ambident oxazolin-5-one anions and N-acylpyridinium cations initially formed under kinetic control, are transformed via the ion pair into the thermodynamically most stable product[28]. Decarboxylative ring opening by the subsequently formed carboxylic acid yields the a-acyl amino ketone[29, 30].
Reactions of isocyanates
Pyridine-catalyzed reactions of isocyanates with carboxylic acids to form amides are found to be strongly accelerated on replacement of pyridine by DMAP. Phenylacetic acid is found to react with phenyl isocyanate in 1,2-dichloroethane at 24°C to give the amide in 66 % yield in less than 5 min; whereas on using the same amount of pyridine only 53% could be isolated after 2h. With triethylamine, only very little is formed besides diphenylurea[31].
Miscellaneous Applications
DMAP has been used in the hardening of epoxy resins with dicyanodiamine, in the transformation of nitriles into thionamides, and in the transfer of silyl groups to tertiary hydroxyl groups[32, 33].
Transfer of Functional Groups
Dimethylarninopyridinium salts are interesting reagents for the transfer of acyl and also cyano and phosphono groups in aqueous medium[34, 35].
- Litvinenko, L. M.; Kirichenko, A. I. Dok. Akad. Nauk SSSR, Ser. Khim. 1967, 176, 97;
- Steglich, W.; Höfle, G. Angew. Chem. 1969, 81, 1001; Angew. Chem. Int. Ed. 1969, 8, 981.
- Scriven, E. F. V.; 4-Dialkylaminopyridines: Super Acylation and Alkylation Catalysts; Chem. Soc. Rev.
- H. Vorbriiggen, Angew. Chem. 84, 348 (1972); Angew. Chem. Int.
- Ed. Engl. 11, 305 (1972). L. Pentimalli, Gazz. Chim. Ital. 94, 902 (1964).
- R. W Taft, C. A. Grob, J . Am. Chem. SOC. 96, 1236 (1974).
- C. W N. Cumper, A. Singleton, J . Chem. SOC. B 1967, 1096.
- A. R. Katritzky, E. Fi! Randall, L. E. Sutton, J. Chem. SOC. 1957 1769.
- H. Lumbroso, J. Barassin, Bull. SOC. Chim. Fr. 1965, 3143.
- C. D. Johnson, I. Roberts, P. G. Taylor, J. Chem. SOC.C hem. Commun 1977, 897.
- M. R. Chakrabarty, C. S. Handloser, M. W Mosher, J. Chem. SOC. Perkin Trans. I1 1973, 938
- Pyridine syntheses, 1st Communication.-2nd Communication: H. Vorbriiggen, J. Kottwitz, K. Krolikiewicz, Chem. Ber., in preparation. This publication gives a complete survey of the various syntheses of DMAP and PPY; H. Vorbriiggen, DOS 2517774 (1975), Schering AG; Chem. Abstr. 86, 55293d (1977).
- W Steglich, G. Hofle, Tetrahedron Lett. 1970, 4727.
- E. Koenigs, H. Friedrich, H. Jurany, Ber. Dtsch. Chem. Ges. 58, 2571 (1925).
- A. C. Satterthwait, W P. Jencks, J. Am. Chem. SOC. 96, 7031 (1974).
- A. G. Burton, R. D. Frampton, C. D. Johnson, A. R. Katritzky, J. Chem. SOC. Perkin Trans. 11 1972, 1940.
- G. B. Barlin, J. A. Benbow, J . Chem. SOC. Perkin Trans. I1 1975,1385.
- J. A. Zoltewicz, J. D. Meyer, Tetrahedron Lett. 1968, 421.
- H. Braun, A. Amann, M. Richter, Angew. Chem. 89,488 (1977); Angew. Chem. lnt. Ed. Engl. 16, 471 (1977).
- B. P. Schaffner, H. Wehrli, Helv. Chim. Acta 55, 2563 (1972).
- 4-Dialkylaminopyridines as acylation catalysts, 4th Communication.-3rd Communication: G. Hofle, W Steglich, Synthesis 1972, 619.
- W Steglich, G. Hofle, Angew. Chem. 81, 1001 (1969); Angew. Chem. Int. Ed. Engl. 8, 981 (1969).
- J. E. McMurry, J . H. Musser, M. S. Ahmad, L. C. Blaszczak, J. Org. Chem. 40, 1829 (1975).
- D. J. Zwanenburg, W A. P. Reynen, Synthesis 1976, 624.
- 1. S. Bindra, A. Grodski, J. Org. Chem. 42, 910 (1977).
- H. Paulsen, H. Hohne, Carhohydr. Res. 58, 484 (1977).
- W Steglich, G. Hiiye, Tetrahedron Lett. 1968, 1619.
- W Steglich, G. HoJe, Chem. Ber. 104, 3644 (1971).
- W Steglich, G. HoJe, Chem. Ber. 102, 1129 (1969).
- G. HiiJe, A. Prox, W Steglich, Chem. Ber. 105, 1718 (1972).
- P. W Henniger, J. K. Van der Drift, DOS 2235390 (1973), Koninklijke Nederlandsche Gist-en Spiritusfabriek N. V.; Chem. Abstr. 78, 124608j
- J. Maurer, DOS 180867O(US Pat. 3530093) (1967), Ciba-Geigy; Chem. Abstr. 71, 1032541 (1969).
- P . C . Sriuastaua, M, Pickering, L. B. Allen, D. G. Streeter, M . 7: Campbell, J. R. Witkowski, R. W Sidwell, R. K. Robins, J. Med. Chem. 20, 256 (1977).
- S. D. Larsen, S. A. Monti, J. Am. Chem. SOC. 99, 8015 (1977).
- M. Wakselman, E. Guibl-Jampel, Tetrahedron Lett. 1970, 1521.
DMAP is a useful highly basic nucleophilic catalyst for a variety of reactions such as esterifications with anhydrides, the Baylis-Hillman reaction, hydrosilylations, tritylation, the Steglich rearrangement.
A highly fluorescent adenosine analogue, which in a dimethoxytrityl, phosphoramidite protected form, can be site-specifically inserted into oligonucleotides through a 3?5?phosphodiester linkage using an automated DNA synthesizer
4-Dimethylaminopyridine is a versatile hypernucleophilic acylation catalyst, it is used to improve the yield, reduce the reaction time, improving relaxation process conditions. Widely used in perfumes, dyes, pigments, pesticides, pharmaceuticals and polymer compounds and other fields. Also used as a catalyst for the synthesis of polyurethane, a curing agent and a blowing catalyst.
In a wide variety of organic syntheses as a catalyst.
4-Dimethylaminopyridine (Valaciclovir EP Impurity G; Valacyclovir USP Related Compound G) is a highly efficient catalyst for acylation reactions.
ChEBI: 4-Dimethylaminopyridine is a dialkylarylamine and a tertiary amino compound.
Valacyclovir Related Compound G, also called as 4-(Dimethylamino)pyridine (DMAP) is an excellent catalyst for acylation of hindered alcohols and in chemical transformations. It is highly nucleophilic in nature.
4-Dimethylaminopyridine (4-DMAP) is readily absorbed through the skin and is highly toxic
by skin absorption. It also causes skin and eye burns. All skin and
eye contact and inhalation should be avoided. Appropriate OSHA/
MSHA-approved respirator, chemical-resistant gloves and impervious or disposable protective clothing should be worn. Work should
be carried out in a chemical fume hood.
Flammability and Explosibility
Non flammable
4-Dimethylaminopyridine can be soluble in methanol, benzene, ethyl acetate, chloroform, methylene chloride, acetone, and acetic acid. Less soluble in ether,
diisopropyl ether, cyclohexane, hexane, and water.
Recrystallise DMAP from toluene [Sadownik et al. J Am Chem Soc 108 7789 1986]. [Beilstein 22 V 112.] § A polystyrene supported version (PS-DMAP) is commercially available.