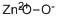Zinc Peroxide, solid and generally similar in its properties to magnesium peroxide. The commercial product is a pale yellow powder containing about 55% ZnO2 and 9% active oxygen. It is stable in dry air but loses its oxygen in moist air and on heating. It is insoluble in water but dissolves in dilute acid, liberating hydrogen peroxide.
White powder containing 45–60%
ZnO2, balance ZnO. Decomposes rapidly above 150C. Decomposes in acids, alcohol, acetone;
insoluble in water but decomposed by it.
Zinc peroxide is used as curing agent, rubber accelerator. has been applied in a variety of settings, from medicine to aesthetics.
Zinc peroxide is used as an accelerator in rubber-compounding, as a curing agent for synthetic elastomers, and as a deodorant for wounds and skin diseases.
Accelerator in rubber compounding; curing agent for synthetic elastomers. Deodorant for wounds and skin diseases.
Slowly decomposed by water.
Zinc peroxide dissolves in dilute acid, liberating hydrogen peroxide [Merck 11th ed. 1989]. Noncombustible but accelerates the burning of combustible material. If the combustible material is finely divided, the mixture may be explosive. Mixtures with combustible material can sometimes be ignited by friction or contact with moisture.The hydrated material (of indefinite composition) explodes at 212°C. Mixtures with aluminum or zinc powder burn brilliantly [Mellor 1940. v. 4, 530].
TOXIC; inhalation, ingestion or contact (skin, eyes) with vapors, dusts or substance may cause severe injury, burns or death. Fire may produce irritating and/or toxic gases. Toxic fumes or dust may accumulate in confined areas (basement, tanks, hopper/tank cars, etc.). Runoff from fire control or dilution water may cause pollution.
May explode from friction, heat or contamination. These substances will accelerate burning when involved in a fire. May ignite combustibles (wood, paper, oil, clothing, etc.). Some will react explosively with hydrocarbons (fuels). Containers may explode when heated. Runoff may create fire or explosion hazard.
Systemic toxicity is
similar to zinc oxide. Flammable when
SYN: ZINC SUPEROXIDE
212.. Can react.iolently with A. and Zn.
Very dangerous, wdl react with water or
steam to produce heat. Vigorous reaction
with reducing materials. When heated to
decomposition it emits toxic fumes of ZnO.
See also PEROXIDES and ZINC
COMPOUNDS.