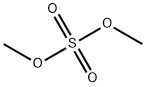
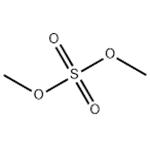
Dimethyl sulfate (chemical formula: (CH3O)2SO2) is an odorless, corrosive, oily liquid which can release toxic fumes during heating. It can be synthesized through the esterification of sulfuric acid with methanol, and alternatively by the distillation of methyl hydrogen sulfate.

In industry, dimethyl sulfate is used as a methylating agent for the manufacture of many organic chemicals. It can be used for methylation of phenols, amines, and thiol. Moreover, it can be used for base sequencing and DNA chain cleavage since it can rupture the imidazole rings present in guanine. It can also be used for protein-DNA interaction analysis. However, its vapor is toxic to eyes and lungs, can do harm to our body. It is a potential carcinogen based on known experimental data.
Dimethyl sulfate is a colorless, oily liquid that is slightly soluble in water. It has a faint, onion-like odor; the odor threshold has not been established. The vapor pressure for dimethyl sulfate is 0.5 mm Hg at 20 °C, and it has a log octanol/water partition coefficient (log Kow ) of 0.032.
Dimethyl sulphate has been used since the beginning of the century as a methylating agent in the preparation of organic chemical products and colouring agents, in the perfume industry, and in other processes. It is a colourless or yellowish liquid of oily consistency which vaporizes at 50℃. and has a slight piquant smell. Both the liquid and the vapour are vesicants and by virtue of this property may be used in warfare.
https://en.wikipedia.org/wiki/Dimethyl_sulfate
https://pubchem.ncbi.nlm.nih.gov/compound/dimethyl_sulfate#section=Top
Dimethyl sulfate is a colorless, oily liquid with a faint, onionlike
odor. It is soluble in water, ether, dioxane, acetone,
benzene, and other aromatic hydrocarbons, miscible with
ethanol, and sparingly soluble in carbon disulfide. It is stable
under normal temperatures and pressures, but hydrolyzes
rapidly in water at or above 18 ℃.
Dimethyl sulfate has been produced commercially since at
least the 1920s. One production method is continuous reaction
of dimethyl ether with sulfur trioxide. In 2009, dimethyl sulfate
was produced by 33 manufacturers worldwide, including 1 in
the United States, 14 in China, 5 in India, 5 in Europe, 6 in East
Asia, and 2 in Mexico, and was available from 44 suppliers,
including 16 US suppliers. There are no data on US imports or
exports of dimethyl sulfate. Reports filed from 1986 through
2002 under the US Environmental Protection Agency’s Toxic
Substances Control Act Inventory Update Rule indicate that US
production plus imports of dimethyl sulfate totaled 10–50
million pounds. The simplest way of synthesizing dimethyl
sulfate is by esterification of sulfuric acid with methanol as
follows:2CH3OH+ H2SO4→(CH3)2SO4 + 2H2O
Dimethyl sulfate is essentially odorless. The specific gravity of this colorless, corrosive, oily liquid is 1.3322 g/cm3. Dimethyl sulfate is soluble in ether, dioxane, acetone, benzene, and other aromatic hydrocarbons. It is sparingly soluble in carbon disulfide and aliphatic hydrocarbons, and only slightly soluble in water (28 g/l at 18 °C) (O'Neil, 2006).
Dimethyl sulfate is a strong alkylating agent and might also react with the carboxylic acid substrate, further reducing the DMS concentration in the mixture. It is used as a methylating agent in themanufacture of many organic compounds,such as, phenols and thiols. Also, it is used inthe manufacture of dyes and perfumes, andas an intermediate for quaternary ammoniumsalts. It was used in the past as a militarypoison.
Dimethyl Sulfate is a diester of methanol and sulfuric acid. Dimethyl Sulfate is commonly used as a reagent for the methylation of phenols, amines, and thiols. Dimethyl Sulfate is an effective and widely used probe for sequence-specific protein-DNA interactions.
ChEBI: Dimethyl sulfate is the dimethyl ester of sulfuric acid. It has a role as an alkylating agent and an immunosuppressive agent.
Dimethyl sulfate is prepared by distillation of an oleum/methanol mixture; technical production using dimethyl ether and SO3 has also been reported (NLM, 2013).
Dimethyl sulfate is a colorless oily liquid, odorless to a faint onion-like odor. Dimethyl sulfate is very toxic by inhalation. Dimethyl sulfate is a combustible liquid and has a flash point of 182°F. Dimethyl sulfate is slightly soluble in water and decomposed by water to give sulfuric acid with evolution of heat. Dimethyl sulfate is corrosive to metals and tissue.
Pure Dimethyl sulfate and concentrated aqueous ammonia react extremely violently with one another, as is the case for tertiary organic bases, [NFPA 491M, 1991]. Dimethyl sulfate ignites in contact with unheated barium chlorite, due to the rapid formation of unstable methyl chlorite. The product of methylating an unnamed material at 110°C was alloyed to remain in a reactor for 80 min. before the reactor exploded. This involved a sulfur ester such as Dimethyl sulfate, [MCA Case History No. 1786].
Dimethyl sulfate is extremely hazardous because of its lack of warning properties and
delayed toxic effects. The vapor of this compound is extremely irritating to the skin, eyes,
and respiratory tract, and contact with the liquid can cause very severe burns to the eyes
and skin. Ingestion of dimethyl sulfate causes burns to the mouth, throat, and
gastrointestinal tract. The effects of overexposure to dimethyl sulfate vapor may be
delayed. After a latent period of 10 hours or more, headache and severe pain to the eyes
upon exposure to light may occur, followed by cough, tightness of the chest, shortness of
breath, difficulty in swallowing and speaking, vomiting, diarrhea, and painful urination.
Fatal pulmonary edema may develop. Systemic effects of dimethyl sulfate include
damage to the liver and kidneys.
Dimethyl sulfate is listed by IARC in Group 2A ("probable human carcinogen") and is
classified as a "select carcinogen" under the criteria of the OSHA Laboratory Standard.
Data indicate that dimethyl sulfate does not specifically harm unborn animals; dimethyl
sulfate is not a developmental toxin. It is a strong alkylating agent and does produce
genetic damage in animals and in bacterial and mammalian cell cultures.
Flammability and Explosibility
Dimethyl sulfate is a combustible liquid (NFPA rating = 2). Toxic dimethyl sulfate vapors are produced in a fire. Carbon dioxide or dry chemical extinguishers should be used to fight dimethyl sulfate fires.
Tumorigen,Mutagen; Reproductive Effector; Human Data; PrimaryIrritant. Industrial use of dimethyl sulfate is based upon itsmethylating properties. It is used as an alkylating agent; inthe manufacture of methyl esters, ethers, and amines; indyes, drugs, perfume, phenol derivatives, and other organicchemicals; as a solvent in the separation of mineral oils; asan intermediate in the manufacture of sodium acetylide synthesis, as well as many pharmaceuticals and pesticides.
If this chemical gets into the eyes, remove anycontact lenses at once and irrigate immediately for at least30 min, occasionally lifting upper and lower lids. Seek medical attention immediately. If this chemical contacts theskin, remove contaminated clothing and wash immediatelywith soap and water. Seek medical attention immediately. Ifthis chemical has been inhaled, remove from exposure,begin rescue breathing (using universal precautions, including resuscitation mask) if breathing has stopped and CPR ifheart action has stopped. Transfer promptly to a medicalfacility. When this chemical has been swallowed, get medical attention. If victim is conscious, administer water ormilk. Do not induce vomiting. Medical observation isrecommended for 2448 h after breathing overexposure, aspulmonary edema may be delayed. As first aid for pulmonary edema, a doctor or authorized paramedic may consideradministering a corticosteroid spray.
Dimethyl sulfate is reasonably anticipated to be a human carcinogenbased on sufficient evidence of carcinogenicity from studies in experimental animals.
Chemical/Physical. Hydrolyzes in water (half-life = 1.2 h) to methanol and sulfuric acid
(Robertson and Sugamori, 1966) via the intermediate methyl sulfuric acid (Du Pont, 1999a)
work with dimethyl sulfate should be conducted in a fume hood to prevent exposure by inhalation, and appropriate impermeable gloves and safety goggles should be worn at all times to prevent skin and eye contact.
This compound requires a shipping label of“POISONOUS/TOXIC MATERIALS, CORROSIVE.” Itfalls in Hazard Class 6.1 and Packing Group I.
Dimethyl sulfate can react violently with ammonium hydroxide, sodium azide, and strong oxidizers.
Excess dimethyl sulfate and waste material containing this substance should be placed in a covered metal container, clearly labeled, and handled according to your institution's waste disposal guidelines.