Tannins and Human Health
General description
“Tannin” was originally coined by Seguin:1. to describe the substances present in vegetable extracts, which are responsible for converting animal skin into leather. In plant extracts, these substances exist as polyphenols of varying molecular sizes and complexities. However, many nonpolyphenolic substances in plants also have all the chemical properties of tannin but have not been tested for their ability to leather hides. 2. There are also other substances that turn hides into leather but are not of plant origin. Tannic acid is a light yellow to tan solid with a faint odor. Sinks and mixes with water. tannin as water-soluble phenolic compounds having a molecular weight between 500 and 3000 D. These polyphenols contain a large number of hydroxyl or other functional groups (1 to 2 per 100 D), and therefore are capable of forming cross-linkages with proteins and other macromolecules. The low-molecular-weight phenolic compounds (molecular weight <500) and those of high molecular weights (molecular weight >3000) are ineffective tanning agents.18 Vegetable tannins can also aggregate with alkaloids, gelatin, and proteins to form precipitation Tannins (commonly referred to as tannic acid) are water-soluble polyphenols that are present in many plant foods. Tannins have been found in a variety of plants utilized as food and feed. These include food grains such as sorghum, millets, barley, dry beans, faba beans, peas, carobs, pigeonpeas, winged beans, and other legumes. Fruits such as apples, bananas, blackberries, cranberries, dates, grapes, hawthornes, peaches, pears, persimmons, plums, raspberries, and strawberries also contain an appreciable quantity of tannins. Likewise, phenolic compounds are present in wines and tea. Forages such as crownvetch, lespedeza, lotus, sainfoin, and trefoil are also reported to contain tannins.[1]
Application and Pharmacology
Tannins (commonly referred to as tannic acid) are water-soluble polyphenols that are present in many plant foods. They have been reported to be responsible for decreases in feed intake, growth rate, feed efficiency, net metabolizable energy, and protein digestibility in experimental animals. Therefore, foods rich in tannins are considered to be of low nutritional value. However, recent findings indicate that the major effect of tannins was not due to their inhibition on food consumption or digestion but rather the decreased efficiency in converting the absorbed nutrients to new body substances. Incidences of certain cancers, such as esophageal cancer, have been reported to be related to consumption of tannins-rich foods such as betel nuts and herbal teas, suggesting that tannins might be carcinogenic. However, other reports indicated that the carcinogenic acitivity of tannins might be related to components associated with tannins rather than tannins themselves. Interestingly, many reports indicated negative association between tea consumption and incidences of cancers. Tea polyphenols and many tannin components were suggested to be anticarcinogenic. Many tannin molecules have also been shown to reduce the mutagenic activity of a number of mutagens. Many carcinogens and/or mutagens produce oxygen-free radicals for interaction with cellular macromolecules. The anticarcinogenic and antimutagenic potentials of tannins may be related to their antioxidative property, which is important in protecting cellular oxidative damage, including lipid peroxidaton. The generation of superoxide radicals was reported to be inhibited by tannins and related compounds. The antimicrobial activities of tannins are well documented. The growth of many fungi, yeasts, bacteria, and viruses was inhibited by tannins. We have also found that tannic acid and propyl gallate, but not gallic acid, were inhibitory to foodborne bacteria, aquatic bacteria, and off-flavor-producing microorganisms. Their antimicrobial properties seemed to be associated with the hydrolysis of ester linkage between gallic acid and polyols hydrolyzed after ripening of many edible fruits. Tannins in these fruits thus serve as a natural defense mechanism against microbial infections. The antimicrobial property of tannic acid can also be used in food processing to increase the shelf-life of certain foods, such as catfish fillets. Tannins have also been reported to exert other physiological effects, such as to accelerate blood clotting, reduce blood pressure, decrease the serum lipid level, produce liver necrosis, and modulate immunoresponses.
Tannic acid could decrease the biofilm formation ability of CRKP. The 3 CRKP isolates showed decreased susceptibilities to imipenem and meropenem,and the MIC were ≥64 μg/mL. Tannic acid could decrease the growth and biofilm formation ability of CRKP. which was further confirmed by scanning electron microscopy. The results of real-time fluorescence quantitative PCR showed that tannic acid could reduce the expression of quorum sensing related gene luxS and lipopolysaccharide related gene wbbM. Tannic acid can inhibit the growth of Escherichia coli and the formation of biofilm, and can be used as a new antibacterial compound. In this study, three crkp strains were collected and one carbapenem sensitive strain KP 13883 was used as the control. The results showed that tannic acid inhibited the growth of crkp and carbapenem sensitive strain KP 13883; Further analysis by 96 well plate crystal violet staining showed that tannic acid could significantly inhibit the biofilm formation of Klebsiella pneumoniae, which was further confirmed by the observation results of scanning electron microscope. Therefore, the mechanism of tannic acid reducing biofilm formation may be to inhibit the growth of bacteria itself.[2] It can be used as food antioxidant. It is generally used after mixing with vitamin E, vitamin C or gallic acid. Tannin can be used as viscosity reducer and filtrate reducer of water-based drilling fluid, and can improve the quality of filter cake. It is also used for tanning raw hides to convert them into leather. Tannin has the activity of reacting with proteins, polysaccharides, alkaloids, microorganisms, enzymes and metal ions, and has the characteristics of antioxidant, free radical capture, bacteriostasis and derivatization. It is more and more widely used in food processing, fruit and vegetable processing, storage, cosmetics, medicine and water treatment. Tannic acid can combine with protein to produce water-insoluble tannic protein, so adding it to shampoo can prevent the dissolution of hair fiber protein and protect hair. Using it in nail products can increase the strength of nails. Gallic tannic acid and isogallic tannic acid have strong free radical scavenging effect. Their effect is similar to that of gallic acid and catechins. They can be used in skin care products to prevent sunscreen and brighten the skin. Tannic acid has astringency and antibacterial properties. It is clinically used to treat local bleeding, protect teeth with toothpaste and prevent gum bleeding.
Synthesis and Degradation
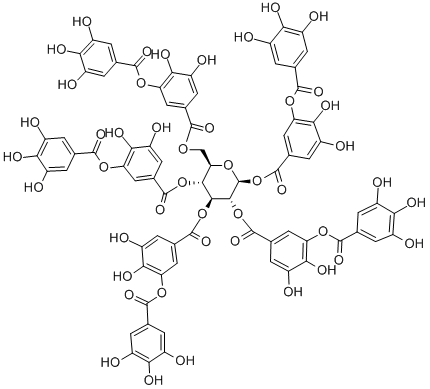
Tannins can be classified into two categories: hydrolyzable and nonhydrolyzable or condensed tannins.19 Hydrolyzable tannins contain a central core of polyhydric alcohol such as glucose, and hydroxyl groups, which are esterified either partially or wholly by gallic acid (gallotannins) or hexahydroxydiphenic acid (ellagitannins). After hydrolysis by acids, bases, or certain enzymes, gallotannins yield glucose and gallic acids. The hexahydroxydiphenic acid of ellagitannins undergoes lactonization to produce ellagic acid (Figure 1).
Figure 1 the degradation of tannic acid
Tannic acid is a strong protein precipitant. In the process of enzymatic preparation of gallic acid, high concentration of tannic acid will seriously inhibit tannic enzyme activity, so it is difficult to directly use tannic acid for industrial production of gallic acid. In order to remove the inhibitory effect of tannic acid on the tannase, a simultaneous extraction-enzymolysis process of the gallic-nut tannic acid was established. The amount of gallic acid could reach 13.9 g/L after 90 min reaction, which is obviously higher than the synchronous extraction-acidolysis process and the stepwise extraction-enzymatic process. The single factor optimization results showed that the optimum extraction-enzymolysis condition for gallic acid is: Material-liquid ratio 1:50, the amount of enzyme 32 U/mL, pH value 6~7, the temperature 50 ℃ and the extraction time was 75 min. Under these conditions, the yield and production rate of gallic acid reached 15.4 g/L and 76.8%, respectively. When conducting multiple feeding experiments under optimal conditions, the concentration of gallic acid could be increased to 34.0 g/L.[3]
Reference
1. "Critical Reviews in Food Science and Nutrition,".
2.Zhou Yanxia, Jin Dongfang: "the effect and mechanism of tannic acid on inhibiting the biofilm formation of Klebsiella pneumoniae", laboratory medicine, No. 10, 2021, pp. 1070-1073.
3.Yang Qiuming, Ru Yi, Weng Huifen, et al. "Study on simultaneous extraction enzymatic hydrolysis process of gallnut tannic acid", food science and technology, 2020, No. 07, pp. 235-241.
);You may like
Lastest Price from Tannic acid manufacturers

US $0.00/Kg/Drum2024-04-28
- CAS:
- 1401-55-4
- Min. Order:
- 1KG
- Purity:
- 60%-98%
- Supply Ability:
- 5000mt/year
US $18.50-14.50/kg2024-04-28
- CAS:
- 1401-55-4
- Min. Order:
- 1kg
- Purity:
- 99%
- Supply Ability:
- 5000kg