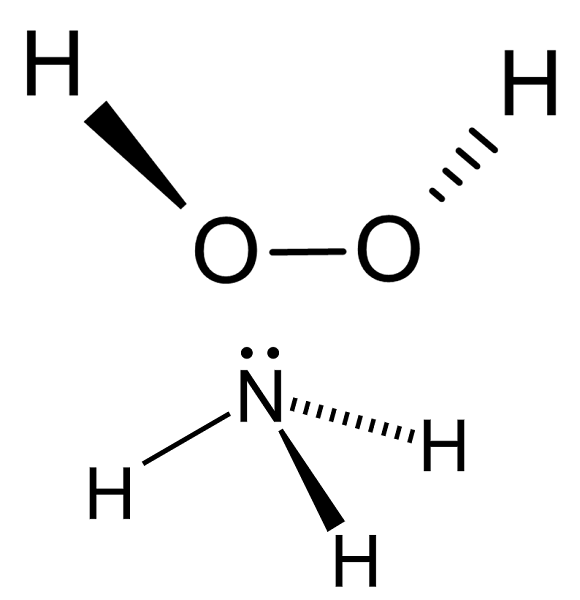
What is the conjugate base of NH3?Is it amphotericital?
Amphotericity of ammonia
One of the most characteristic properties of ammonia is its basicity. Ammonia is considered to be a weak base. It combines with acids to form ammonium salts; thus, with hydrochloric acid, it forms ammonium chloride (sal ammoniac); with nitric acid, ammonium nitrate, etc. Perfectly dry ammonia gas will not combine with perfectly dry hydrogen chloride gas; moisture is necessary to bring about the reaction.
As a demonstration experiment under air with ambient moisture, opened bottles of concentrated ammonia and hydrochloric acid solutions produce a cloud of ammonium chloride, which seems to appear 'out of nothing' as the salt aerosol forms where the two diffusing clouds of reagents meet between the two bottles.
NH3 + HCl → [NH4]Cl
The salts produced by the action of ammonia on acids are known as the ammonium salts and all contain the ammonium ion ([NH4]+).
Although ammonia is well known as a weak base, it can also act as an extremely weak acid. It is a protic substance and is capable of the formation of amides (which contain the NH−2ion). For example, lithium dissolves in liquid ammonia to give a blue solution (solvated electron) of lithium amide:
2 Li + 2 NH3 → 2 LiNH2 + H2
Conjugate base of ammonium
The conjugate base of NH3 is NH2- which is known as the azide anion.
Production of ammonium
About 80% of the ammonia produced in industry is used in agriculture as fertilizer. Ammonia is also used as a refrigerant gas, to purify water supplies, and in the manufacture of plastics, explosives, fabrics, pesticides, dyes, and other chemicals. It is found in many household and industrial-strength cleaning solutions. Cleaning solutions for industrial use contain higher concentrations of ammonia and can quickly cause irritation and burns.
);You may like
Related articles And Qustion
Lastest Price from Ammonia manufacturers

US $2.00/kg2024-02-13
- CAS:
- 7664-41-7
- Min. Order:
- 10000kg
- Purity:
- 99%
- Supply Ability:
- 10000000