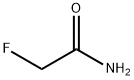
Description
Fluoroacetamide is an odourless, tasteless, white, crystalline water soluble. It is a fluorinated
amide and reacts with azo and diazo compounds to generate toxic gases. Flammable
gases are formed by the reaction of organic amides/imides with strong reducing agents.
Fluoroacetamide is used as a rodenticide and insecticide to control rabbits but is dangerous
for other species of pests, farm livestock, and humans. Fluoroacetamide is a noncombustible
substance itself and does not burn but may decompose upon heating to
produce irritating, corrosive, and/or toxic fumes. On decomposition, fluoroacetamide
releases nitrogen oxides, carbon monoxide, irritating and toxic fumes and gases, carbon
dioxide, HF gas, and nitrogen gas.
Chemical Properties
crystals
Chemical Properties
Fluoroacetamide is a colorless, crystalline
solid.
Uses
Rodenticide. Insecticide proposed mainly for use on fruits to combat scale insects, aphids, and mites.
Definition
ChEBI: Acetamide substituted at C-2 by a fluorine atom.
Production Methods
Fluoroacetamide is produced by reaction of α-chloroacetamide
with potassium fluoride in tetrachloroethylene
at elevated temperature.
General Description
Colorless crystalline powder. Used as a rodenticide. Highly toxic.
Air & Water Reactions
Very soluble in water [Farm Chemicals]
Reactivity Profile
FLUOROACETAMIDE is a fluorinated amide. Organic amides/imides react with azo and diazo compounds to generate toxic gases. Flammable gases are formed by the reaction of organic amides/imides with strong reducing agents. Amides are very weak bases (weaker than water). Imides are less basic yet and in fact react with strong bases to form salts. That is, they can react as acids. Mixing amides with dehydrating agents such as P2O5 or SOCl2 generates the corresponding nitrile. The combustion of these compounds generates mixed oxides of nitrogen (NOx).
Hazard
Extremely toxic; poison; mutagen.
Health Hazard
FLUOROACETAMIDE is super toxic; probable oral lethal dose in humans is less than 5 mg/kg, or a taste (less than 7 drops) for a 150-lb. person. Chemically inhibits oxygen metabolism by cells with critical damage occurring to the heart, brain, and lungs resulting in heart failure, respiratory arrest, convulsions, and death.
Fire Hazard
Emits very toxic fumes of fluorine containing compounds and nitrogen oxides when heated to decomposition. Avoid decomposing heat.
Safety Profile
A human poison by an
unspecified route. Poison experimentally by
ingestion, skin contact, intraperitoneal,
subcutaneous, and intravenous routes.
Human systemic effects by unspecified
route: convulsions, coma, nausea and
vomiting. Experimental reproductive effects.
Mutation data reported. Used as an
insecticide and rodenticide. When heated to
decomposition it emits very toxic fumes of
Fand NOx. See also FLUORIDES.
Potential Exposure
This material is an organofluorine
rodenticide; insecticide proposed mainly for use on fruits to
combat scale insects, aphids, and mites. Use is largely
restricted to licensed pest control operators.
storage
Color Code—Blue: Health Hazard/Poison: Storein a secure poison location. Prior to working with thischemical you should be trained on its proper handling andstorage. Store in tightly closed containers in a cool, wellventilated area.
Shipping
UN2811 Toxic solids, organic, n.o.s., Hazard
Class: 6.1; Labels: 6.1-Poisonous materials, Technical
Name Required.
Purification Methods
Crystallise fluoroacetamide from chloroform and dry it in a vacuum. [Beilstein 2 IV 454.]
Incompatibilities
Incompatible with oxidizers (chlorates,
nitrates, peroxides, permanganates, perchlorates, chlorine,
bromine, fluorine, etc.); contact may cause fires or explo sions. Keep away from alkaline materials, strong bases,
strong acids, oxoacids, epoxides.
Waste Disposal
Consult with environmental
regulatory agencies for guidance on acceptable disposal
practices. Generators of waste containing this contaminant
(≥100 kg/mo) must conform with EPA regulations govern ing storage, transportation, treatment, and waste disposal.